Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(C\%=\dfrac{30}{170}.100\%=17,647\%\)
\(V_{\text{dd}}=\left(30+170\right)1,1=220ml\)
\(n_{NaCl}=\dfrac{30}{58,5}=0,513mol\)
\(C_M=\dfrac{0,513}{0,22}=0,696M\)
\(C\%_{NaCl}=\dfrac{30}{170+30}.100\%=15\%\\ C_M=C\%.\dfrac{10D}{M}=10.\dfrac{10.1,1}{58,5}=1,88M\)

\(n_{Na_2CO_3.10H_2O}=\dfrac{28,6}{286}=0,1\left(mol\right)\)
=> nNa2CO3 = 0,1(mol)
=> \(C_M=\dfrac{0,1}{0,2}=0,5M\)
mdd sau pư = 1,05.200 = 210 (g)
=> \(C\%=\dfrac{0,1.106}{210}.100\%=5,05\%\)

\(n_{NaOH}=\dfrac{4}{40}=0,1\left(mol\right)\\ C_{M_{NaOH}}=\dfrac{0,1}{0,4}=0,25\left(M\right)\)
n NaOH= \(\dfrac{4}{40}\)=0,1(mol)
C MnaoH=\(\dfrac{0,1}{0,4}\)=0,25(M)

a)
\(n_{FeSO_4.7H_2O}=\dfrac{41,7}{278}=0,15\left(mol\right)\)
=> \(n_{FeSO_4}=0,15\left(mol\right)\)
=> \(m_{FeSO_4}=0,15.152=22,8\left(g\right)\)
b) mdd sau pha trộn = 41,7 + 207 = 248,7 (g)
c) \(C\%=\dfrac{22,8}{248,7}.100\%=9,168\%\)
\(V_{dd}=\dfrac{248,7}{1,023}=243,1085\left(ml\right)=0,2431085\left(l\right)\)
\(C_M=\dfrac{0,15}{0,2431085}=0,617M\)

a)
C% CuSO4 = 16/(16 + 184) .100% = 8%
b)
n NaOH = 20/40 = 0,5(mol)
CM NaOH = 0,5/4 = 0,125M

a) \(C\%=\dfrac{m_{KCl}}{m_{ddKCl}}.100\%=\dfrac{10}{300}.100\%\approx3,3\%\)
b) Đổi: \(1500ml=1,5l\)
\(C_{MCuSO_4}=\dfrac{n}{V}=\dfrac{3}{1,5}=2M\)
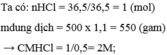
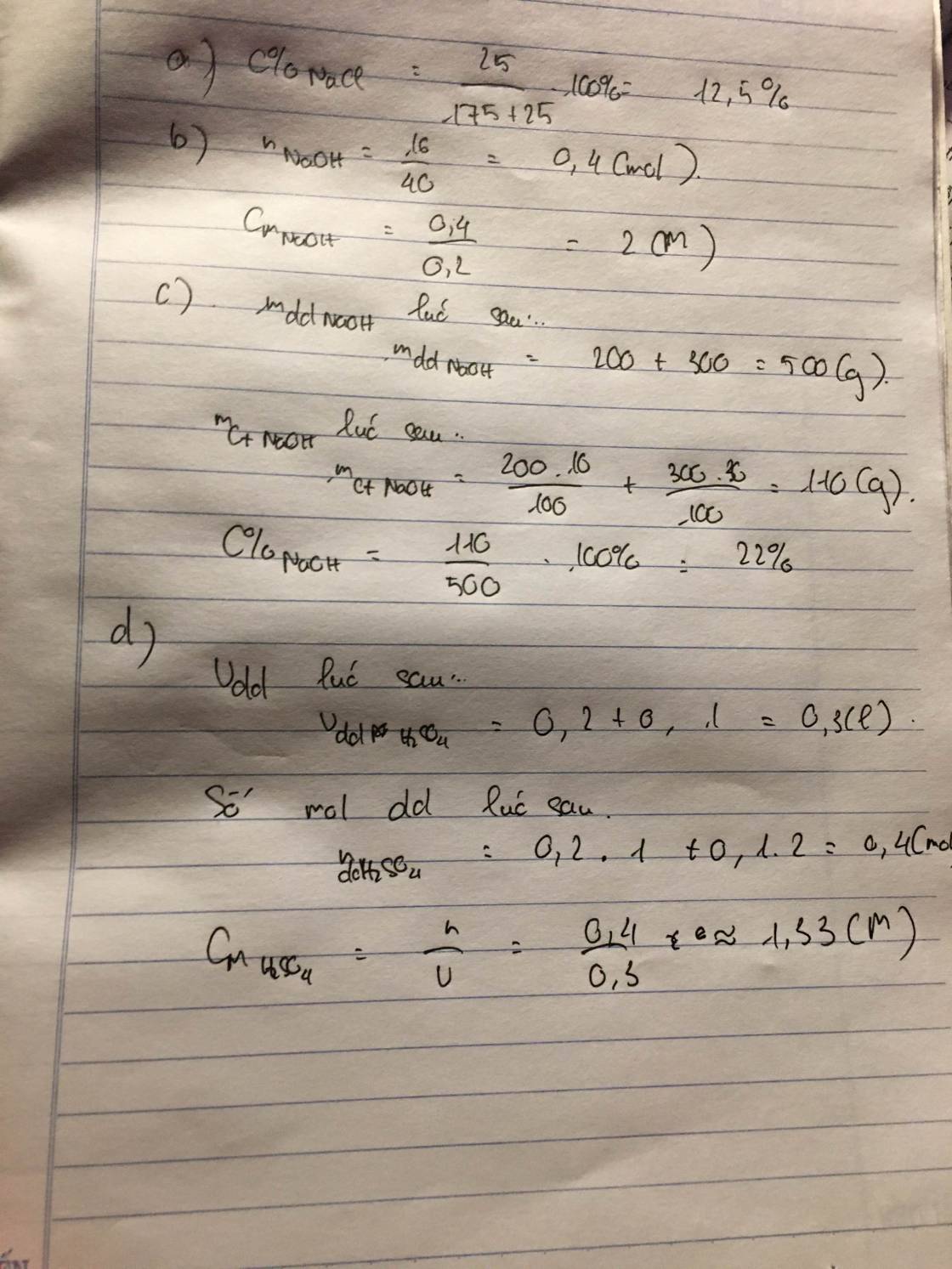
Ta có: \(n_{Na_2CO_3}=n_{Na_2CO_3.10H_2O}=\dfrac{38,61}{286}=0,135\left(mol\right)\)
m dd sau pư = 38,61 + 256 = 294,61 (g)
\(\Rightarrow C\%_{Na_2CO_3}=\dfrac{0,135.106}{294,61}.100\%\approx4,86\%\)
Có: \(V_{ddsaupư}=\dfrac{294,61}{1,156}\approx254,85\left(ml\right)\approx0,255\left(l\right)\)
\(\Rightarrow C_{M_{Na_2CO_3}}=\dfrac{0,135}{0,255}\approx0,53M\)
Bạn tham khảo nhé!
Gọi số mol của Na2CO3 là a (mol) \(\Rightarrow n_{H_2O\left(phân.tử\right)}=10a\left(mol\right)\)
\(\Rightarrow106a+18\cdot10a=38,61\) \(\Leftrightarrow a=0,135\left(mol\right)\)
\(\Rightarrow C\%_{Na_2CO_3}=\dfrac{0,135\cdot106}{38,61+256}\cdot100\%\approx4,86\%\)
Mặt khác: \(V_{ddNa_2CO_3}=\dfrac{38,61+256}{1,156}\approx254,41\left(ml\right)\) \(\Rightarrow C_{M_{Na_2CO_3}}=\dfrac{0,135}{0,25441}\approx0,53\left(M\right)\)