Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{H_2}=\dfrac{1.12}{22.4}=0.05\left(mol\right)\)
\(2A+2nH_2O\rightarrow2A\left(OH\right)_n+nH_2\)
\(\dfrac{0.1}{n}........................0.05\)
\(M_A=\dfrac{3.9}{\dfrac{0.1}{n}}=39n\)
Với : \(n=1\rightarrow A=39\)
\(A:K\)
\(m_{KOH}=0.1\cdot56=5.6\left(g\right)\)
\(m_{ddX}=3.9+46.2-0.05\cdot2=50\left(g\right)\)
\(C\%_{KOH}=\dfrac{5.6}{50}\cdot100\%=11.2\%\)
\(b.\)
\(K_2O+H_2O\rightarrow2KOH\)
\(0.1....................0.2\)
\(m_{KOH}=0.2\cdot56=11.2\left(g\right)\)
\(m_{dd_X}=\dfrac{11.2}{28\%\%}=40\left(g\right)\)

\(\text{2R+2H2O->2ROH+H2}\)
\(\text{ROH+HCl->RCl+H2O}\)
nROH=nHCl=0,2.1=0,2(mol)
nH2=\(\frac{nROH}{2}\)=\(\frac{0,2}{2}\)=0,1(mol)
V=0,1.22,4=2,24(l)
\(\text{nR=nROH=0,2(mol)}\)
\(\text{=>MR=7,8/0,2=39(g)}\)
R là Kali(K)
\(\text{2KOH+CuSO4->Cu(OH)2+K2SO4}\)
\(\text{nCuSO4=0,3x0,5=0,15(mol)}\)
=>nCuSO4 dư=0,15-0,1=0,05(mol)
m kết tủa =0,1.98=9,8(g)
\(\left\{{}\begin{matrix}\text{CMK2SO4=0,1/0,5=0,2(M)}\\\text{CMCuSO4=0,05/0,5=0,1(M)}\end{matrix}\right.\)

`1)`
`n_{Al}={2,7}/{27}=0,1(mol)`
`2Al+3H_2SO_4->Al_2(SO_4)_3+3H_2`
`0,1->0,15->0,05->0,15(mol)`
`V_{dd\ H_2SO_4}={0,15}/1=0,15(l)=150(ml)`
`->V=150`
`V'=V_{H_2}=0,15.22,4=3,36(l)`
`C_{M\ X}=C_{M\ Al_2(SO_4)_3}={0,05}/{0,15}=1/3M`
`2)`
`n_{Fe}={2,8}/{56}=0,05(mol)`
`Fe+2HCl->FeCl_2+H_2`
`0,05->0,1->0,05->0,05(mol)`
`V_{dd\ HCl}={0,1}/1=0,1(l)=100(ml)`
`->V=100`
`V_{H_2}=0,05.22,4=1,12(l)`
`C_{M\ FeCl_2}={0,05}/{0,1}=0,5M`

\(Zn+2HCl->ZnCl_2+H_2\\ Fe+2HCl->FeCl_2+H_2\\n_{Zn}=a;n_{Fe}=b\\ 65a+56b=2,1\\ 2\left(a+b\right)=0,4\\ a=-1,011;b=1,211\)
Đáp số ra số âm, không thoả mãn điều kiện thực tế a, b > 0.

Gọi: CT của 2 muối clorua : ACl2 ( x mol)
BCl2 (3x mol)
mhh= x (A+71) + 3x (B + 71)= 5.94g
<=> x (A + 71 + 3B + 213)= 5.94
<=> x(A+3B+284)= 5.94 (1)
nAgCl= 17.22/143.5=0.12 mol
ACl2 + 2AgNO3 --> A(NO3)2 + 2AgCl
x____________________________2x
BCl2 + 2AgNO3 --> B(NO3)2 + 2AgCl
3x__________________________6x
nAgCl= 2x + 6x = 0.12
=> x = 0.015
Thay x vào (1)
<=> A + 3B = 112 (2)
Mặt khác:
MA/ MB = 5/ 3
<=> 3A = 5B
<=> 3A - 5B= 0 (3)
Giải (2) và (3):
A= 40 (Ca)
B= 24 (Mg)
CMCaCl2= 0.015/0.1=0.15 M
CMMgCl2= 0.045/0.1=0.45M
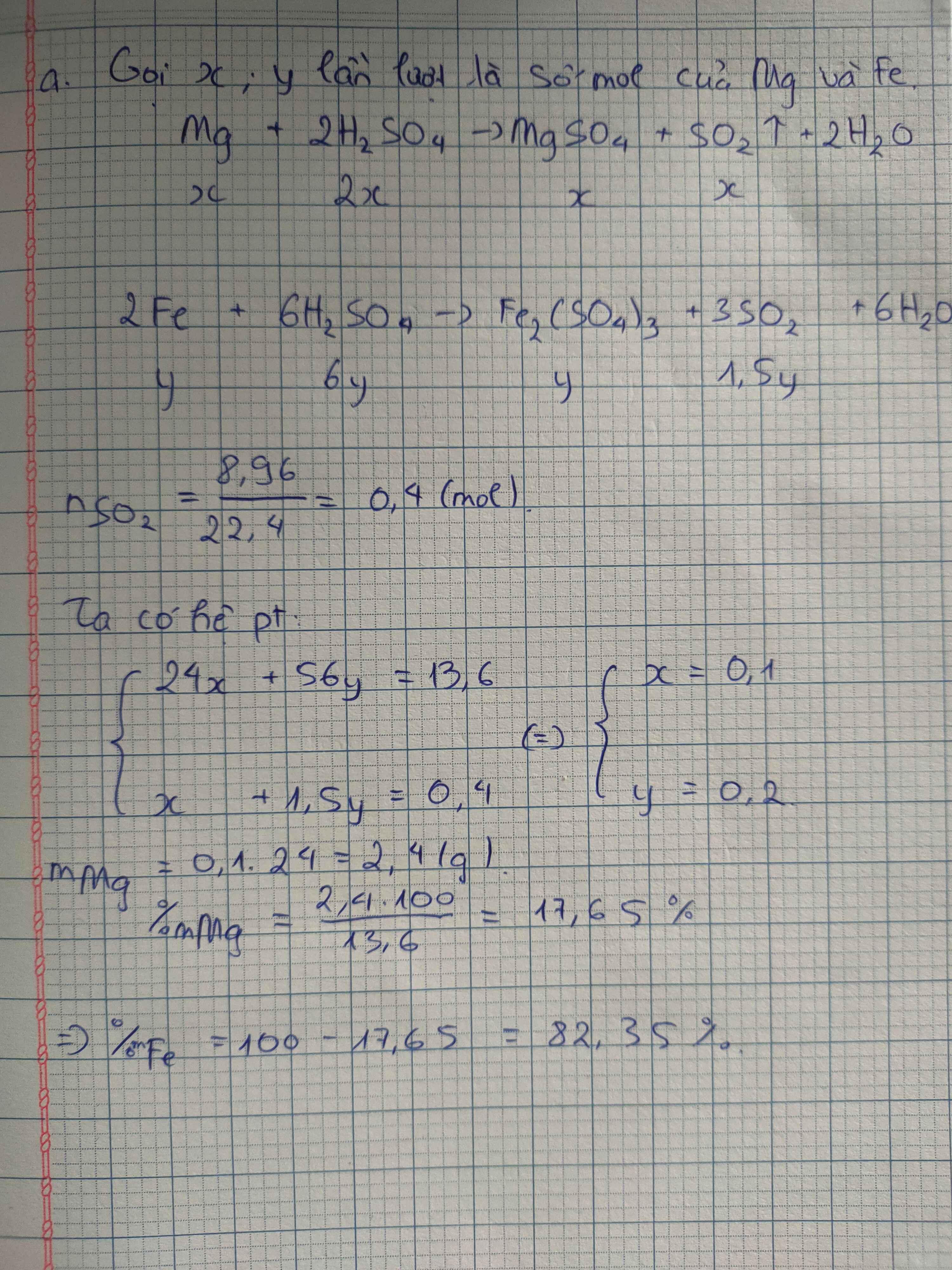


b/
\(2R+6H_2SO_4\rightarrow R_2\left(SO_4\right)_3+3SO_2+6H_2O\)
0,15 0,45 0,075 0,225 (mol)
n\(_{SO_2}=\)\(\dfrac{5,04}{22,4}\)=0,225(mol)
M\(_R=\dfrac{4,05}{0,15}=27\left(g\right)\)
\(\rightarrow\)nhôm(Al)
b/
m\(_{H_2SO_{4\left(cần\right)}}\)=\(\dfrac{0,45.98.100}{80}=55,125\left(g\right)\)
\(m_{H_2SO_{4\left(lấy\right)}}=55,125.105\%=57,88125\left(g\right)\)
m\(_{dd}\)=4,05+57,88125=61,93125(g)
m\(_{Al_2\left(SO_4\right)_3=0,075.342=25,65\left(g\right)}\)
C%\(_{Al_2\left(SO_4\right)_3}=\dfrac{25,65}{\text{61,93125}}.100\approx41,4\%\)
C%\(_{H_2SO_{4\left(dư\right)}}=\dfrac{55,125.5}{\text{61,93125}.100}.100\approx4,45\%\)