Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{CaCO_3}=\dfrac{25}{100}=0.25\left(mol\right)\)
\(Ca\left(OH\right)_2+CO_2\rightarrow CaCO_3+H_2O\)
\(0.25...........0.25...........0.25\)
\(C_{M_{Ca\left(OH\right)_2}}=\dfrac{0.25}{0.1}=2.5\left(M\right)\)
\(CH_4+2O_2\underrightarrow{t^0}CO_2+2H_2O\)
\(0.25..............0.25\)
\(V_{CH_4}=0.25\cdot22.4=5.6\left(l\right)\)
a) nCaCO3=0,25(mol)
CH4 + 2 O2 -to-> CO2 + 2 H2O
0,25<------------------0,25(mol)
CO2 + Ca(OH)2 -> CaCO3 + H2O
0,25<------0,25-----------0,25(mol)
b) CMddCa(OH)2= 0,25/0,1= 2,5(M)
b) V(CH4,đktc)=0,25.22,4=5,6(l)

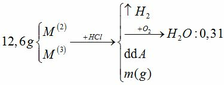
Đồng nhất dữ kiện để thuận lợi cho tính toán, bằng cách nhân đôi khối lượng H2O.
a.
BTNT H: nH2 = nH2O = 0,31 mol
=> nHCl = 0,62mol
BTKL: m kim loại + mHCl = mA + mB => 12,6 + 36,5 . 0,62 = m + 2 . 0,31 => m = 34,61g
b.
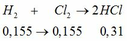
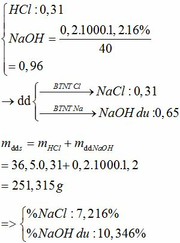

a) \(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
PTHH: 2Al + 3H2SO4 --> Al2(SO4)3 + 3H2
______0,2---->0,3------------>0,1------>0,3______(mol)
=> VH2 = 0,3.22,4= 6,72(l)
b) \(C_{M\left(H_2SO_4\right)}=\dfrac{0,3}{0,1}=3M\)
\(C_{M\left(Al_2\left(SO_4\right)_3\right)}=\dfrac{0,1}{0,1}=1M\)
Câu 3:
\(n_{H_2}=\dfrac{6,72}{22,4}=0,3(mol)\\ PTHH:Mg+2HCl\to MgCl_2+H_2\\ MgO+2HCl\to MgCl_2+H_2O\\ \Rightarrow n_{Mg}=n_{H_2}=0,3(mol)\\ \Rightarrow \%_{Mg}=\dfrac{0,3.24}{15,2}.100\%=47,37\%\\ \Rightarrow \%_{MgO}=100\%-47,37\%=52,63\%\)
\(n_{MgO}=\dfrac{15,2-0,3.24}{40}=0,2(mol)\\ \Rightarrow \Sigma n_{HCl}=0,3.2+0,2.2=1(mol)\\ \Rightarrow m_{dd_{HCl}}=\dfrac{1.36,5}{10\%}=365(g)\\ \Sigma n_{MgCl_2}=0,2+0,3=0,5(mol)\\ \Rightarrow C\%_{MgCl_2}=\dfrac{0,5.95}{15,2+365}.100\%=12,49\%\)
\(PTHH:Mg+2H_2SO_{4(đ)}\to MgSO_4+2H_2O+SO_2\uparrow\\ MgO+H_2SO_4\to MgSO_4+H_2O\\ \Rightarrow n_{SO_2}=n_{Mg}=0,3(mol)\\ \Rightarrow V_{SO_2}=0,3.22,4=6,72(l)\)

a) \(n_{Al}=\dfrac{2,7}{27}=0,1\left(mol\right)\)
PTHH: 4Al + 3O2 ---to→ 2Al2O3
Mol: 0,1 0,075 0,05
\(V_{O_2}=0,075.22,4=1,68\left(l\right)\)
b) \(m_{Al_2O_3}=0,05.102=5,1\left(g\right)\)
c)
PTHH: Al2O3 + 6HCl → 2AlCl3 + 3H2O
Mol: 0,05 0,3 0,1
\(m_{ddHCl}=\dfrac{0,3.36,5.100}{7,3}=150\left(g\right)\)

a, \(n_{C_2H_4}=\dfrac{1,12}{22,4}=0,05\left(mol\right)\)
PT: \(C_2H_4+3O_2\underrightarrow{t^o}2CO_2+2H_2O\)
\(CO_2+Ca\left(OH\right)_2\rightarrow CaCO_{3\downarrow}+H_2O\)
Theo PT: \(n_{CaCO_3}=n_{CO_2}=2n_{C_2H_4}=0,1\left(mol\right)\)
\(\Rightarrow m_{CaCO_3}=0,1.100=10\left(g\right)\)
b, Theo PT: \(n_{O_2}=3n_{C_2H_4}=0,15\left(mol\right)\Rightarrow V_{O_2}=0,15.22,4=3,36\left(l\right)\)
\(\Rightarrow V_{kk}=\dfrac{V_{O_2}}{20\%}=16,8\left(l\right)\)
PT: \(2Al+3Cl_2\underrightarrow{t^o}2AlCl_3\)
\(AlCl_3+3AgNO_3\rightarrow Al\left(NO_3\right)_3+3AgCl\)
Ta có: \(n_{AgNO_3}=0,1.0,6=0,06\left(mol\right)\)
Theo PT: \(n_{Al}=n_{AlCl_3}=\dfrac{1}{3}n_{AgNO_3}=0,02\left(mol\right)\)
⇒ m = mAl = 0,02.27 = 0,54 (g)
\(C_{M_{AlCl_3}}=\dfrac{0,02}{0,15}=\dfrac{2}{15}\left(M\right)\)