Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{C_2H_2}=\dfrac{2,24}{22,4}=0,1mol\)
\(2C_2H_2+5O_2\rightarrow\left(t^o\right)4CO_2+2H_2O\)
0,1 0,25 ( mol )
\(V_{kk}=0,25.22,4.5=28l\)

\(n_{Mg}=\dfrac{7,2}{24}=0,3\left(mol\right)\\ n_{Cl_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\\ Mg+Cl_2\rightarrow\left(t^o\right)MgCl_2\\ a,Vì:\dfrac{0,2}{1}< \dfrac{0,3}{1}\Rightarrow Mgdư\\n_{Mg\left(p.ứ\right)}=n_{MgCl_2}=n_{Cl_2}=0,2\left(mol\right)\\ \Rightarrow n_{Mg\left(dư\right)}=0,3-0,2=0,1\left(mol\right)\\ m_{Mg\left(dư\right)}=0,1.24=2,4\left(g\right)\\ b,m_{MgCl_2}=0,2.95=19\left(g\right)\)
\(n_{Mg}=\dfrac{m}{M}=\dfrac{7,2}{24}=0,3\) (mol)
\(n_{Cl_2}=\dfrac{V}{22,4}=\dfrac{4,48}{22,4}=0,2\)(mol)
PTHH : Mg + Cl2 ---> MgCl2
1 : 1 : 1
Dễ thấy : \(\dfrac{n_{Mg}}{1}>\dfrac{n_{Cl_2}}{1}\)
=> Mg dư 0,1 mol
=> \(m_{Mg}=n.M=0,1.24=2,4\left(g\right)\)
=> \(n_{MgCl_2}=0,2\left(mol\right)\) => \(m_{MgCl_2}=n.M=0,2.\left(24+71\right)=19\left(g\right)\)
\(n_{Mg}=\dfrac{m}{M}=\dfrac{7,2}{24}=0,3\) (mol)
\(n_{Cl_2}=\dfrac{V}{22,4}=\dfrac{4,48}{22,4}=0,2\)(mol)
PTHH : Mg + Cl2 ---> MgCl2
1 : 1 : 1
Dễ thấy : \(\dfrac{n_{Mg}}{1}>\dfrac{n_{Cl_2}}{1}\)
=> Mg dư 0,1 mol
=> \(m_{Mg}=n.M=0,1.24=2,4\left(g\right)\)
=> \(n_{MgCl_2}=0,2\left(mol\right)\) => \(m_{MgCl_2}=n.M=0,2.\left(24+71\right)=19\left(g\right)\)

\(n_{CaCO_3}=\dfrac{200}{100}=2\left(mol\right)\)
PTHH: CaCO3 --to--> CaO + CO2
_______a-------------->a----->a
=> 100(2-a) + 56a = 129,6
=> a = 1,6 (mol)
=> VCO2 = 1,6 .22,4 = 35,84(l)

a) \(n_{CH_4}=\dfrac{11,2}{22,4}=0,5\left(mol\right)\)
PTHH: CH4 + 2O2 --to--> CO2 + 2H2O
0,5--->1
=> VO2 = 1.22,4 = 22,4 (l)
b) Vkk = 22,4.5 = 112 (l)

Gọi số mol CH4, C2H6 là a, b
=> a+b = \(\dfrac{5,6}{22,4}=0,25\)
Có \(\dfrac{16a+30b}{a+b}=0,6.29=17,4\)
=> a = 0,225; b = 0,025
PTHH: CH4 + 2O2 --to--> CO2 + 2H2O
____0,225->0,45------->0,225->0,45
2C2H6 + 7O2 --to--> 4CO2 + 6H2O
0,025->0,0875----->0,05-->0,075
=> VO2 = (0,45+0,0875).22,4 = 12,04 (l)
mCO2 = (0,225 + 0,05).44 = 12,1(g)
mH2O = (0,45+ 0,075).18 = 9,45(g)

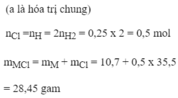
a)
$n_{O_2} = \dfrac{2,24}{22,4} = 0,1(mol)$
$m_{O_2} = 0,1.32 = 3,2(gam)$
b)
$n_{Cl_2} = \dfrac{7,1}{71} = 0,1(mol)$
$V_{Cl_2} = 0,1.22,4 = 2,24(lít)$