Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a, \(n_{HCl}=0,2.0,1=0,02\left(mol\right)=n_{H^+}=n_{Cl^-}\)
\(n_{H_2SO_4}=0,2.0,15=0,03\left(mol\right)=n_{SO_4^{2-}}\) \(\Rightarrow n_{H^+}=2n_{H_2SO_4}=0,06\left(mol\right)\)
\(\Rightarrow\Sigma n_{H^+}=0,02+0,06=0,08\left(mol\right)\)
\(n_{Ba\left(OH\right)_2}=0,3.0,05=0,015\left(mol\right)=n_{Ba^{2+}}\)
\(\Rightarrow n_{OH^-}=2n_{Ba\left(OH\right)_2}=0,03\left(mol\right)\)
\(H^++OH^-\rightarrow H_2O\)
0,03___0,03 (mol) ⇒ nH+ dư = 0,05 (mol)
\(Ba^{2+}+SO_4^{2-}\rightarrow BaSO_4\)
0,015___0,015______0,015 (mol) ⇒ nSO42- dư = 0,015 (mol)
⇒ m = mBaSO4 = 0,015.233 = 3,495 (g)
\(\left[Cl^-\right]=\dfrac{0,02}{0,2+0,3}=0,04\left(M\right)\)
\(\left[H^+\right]=\dfrac{0,05}{0,2+0,3}=0,1\left(M\right)\)
\(\left[SO_4^{2-}\right]=\dfrac{0,015}{0,2+0,3}=0,03\left(M\right)\)
b, pH = -log[H+] = 1

Chọn C
pH = 11 → [ OH - ] = 10 - 3 (M)
pH = 12 → [ OH - ] = 10 - 2 (M)
Tổng số mol OH - có trong dung dịch X là: n = 0 , 1 . 10 - 3 + 0 , 05 . 10 - 2 = 6 . 10 - 4 (mol)
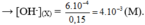

\(n_{NaOH}=0,1.0,2=0,02\left(mol\right)\\ n_{HCl}=0,3.0,1=0,03\left(mol\right)\\ NaOH+HCl\rightarrow NaCl+H_2O\\ Vì:\dfrac{0,02}{1}< \dfrac{0,03}{1}\Rightarrow HCldư\\ n_{HCl\left(dư\right)}=0,03-0,02=0,01\left(mol\right)\\ \left[H^+\right]=\left[HCl_{dư}\right]=\dfrac{0,01}{0,1+0,1}=0,05\left(M\right)\\ \Rightarrow D\)

\(n_{NaOH}=0.25\cdot2=0.5\left(mol\right)\)
\(n_{H_2SO_4}=0.25\cdot1=0.25\left(mol\right)\)
\(2NaOH+H_2SO_4\rightarrow Na_2SO_4+H_2O\)
\(0.5..............0.25................0.25\)
\(\left[Na^+\right]=\dfrac{0.25\cdot2}{0.25+0.25}=1\left(M\right)\)
\(\left[SO_4^{2-}\right]=\dfrac{0.25}{0.25+0.25}=0.5\left(M\right)\)

a, \(n_{HCl}=0,1.0,2=0,02\left(mol\right)=n_{H^+}=n_{Cl^-}\)
\(n_{H_2SO_4}=0,1.0,2=0,02\left(mol\right)=n_{SO_4^{2-}}\) \(\Rightarrow n_{H^+}=2n_{H_2SO_4}=0,04\left(mol\right)\)
\(n_{NaOH}=0,3.0,4=0,12\left(mol\right)=n_{Na^+}=n_{OH^-}\)
\(\Rightarrow\sum n_{H^+}=0,02+0,04=0,06\left(mol\right)\)
\(H^++OH^-\rightarrow H_2O\)
0,06__0,06 (mol)
⇒ nOH- dư = 0,12 - 0,06 = 0,06 (mol)
\(\Rightarrow\left\{{}\begin{matrix}\left[Cl^-\right]=\dfrac{0,02}{0,1+0,3}=0,05\left(M\right)\\\left[SO_4^{2-}\right]=\dfrac{0,02}{0,1+0,3}=0,05\left(M\right)\\\left[Na^+\right]=\dfrac{0,12}{0,1+0,3}=0,3\left(M\right)\\\left[OH^-\right]=\dfrac{0,06}{0,1+0,3}=0,15\left(M\right)\end{matrix}\right.\)
b, pH = 14 - (-log[OH-]) ≃ 13,176

a, \(n_{H^+}=n_{OH^-}=9.10^{-3}\left(mol\right)\Rightarrow C_{M\left(H_2SO_4\right)}=\dfrac{\dfrac{9.10^{-3}}{2}}{0,05}=0,09M\)
b, \(\left[SO_4^{2-}\right]=\dfrac{4,5.10^{-3}}{0,05+0,15}=0,6M\)
\(\left[Na^+\right]=\dfrac{0,15.0,06}{0,05+0,15}=0,045M\)
\(\left[H^+\right]=\left[OH^-\right]=\dfrac{9.10^{-3}}{0,05+0,15}=0,045M\)

a, \(\left[Na^+\right]=0,1\)
\(\left[K^+\right]=0,1\)
\(\left[OH^-\right]=0,2\)
\(\left[SO_4^{2-}\right]=0,2\)
\(\left[H^+\right]=0,4\)
b, \(n_{H^+}=0,1.0,4=0,04\left(mol\right)\)
\(n_{OH^-}=0,1.0,2=0,02\left(mol\right)\)
\(H^++OH^-\rightarrow H_2O\)
\(\Rightarrow n_{H^+dư}=0,02\left(mol\right)\)
\(\Rightarrow\left[H^+\right]=\dfrac{0,02}{200}=10^{-4}\)
\(\Rightarrow pH=4\)

$n_{NaOH} = n_{KOH} = 0,1.0,1 = 0,01(mol)$
$n_{H_2SO_4} = 0,02(mol)$
OH- + H+ → H2O
Bđ : 0,01...0,04..................(mol)
Pư : 0,01...0,01...................(mol)
Sau pư : 0......0,03...................(mol)
$V_{dd} = 0,1 + 0,1 = 0,2(lít)$
Vậy :
$[K^+] = [Na^+] = \dfrac{0,01}{0,2} = 0,05M$
$[H^+] = \dfrac{0,03}{0,2} = 0,15M$
$[SO_4^{2-}] = \dfrac{0,02}{0,2} = 0,1M$
b)
$pH = -log(0,15) = 0,824$
\(n_{NaOH}=0,15.0,6=0,09\left(mol\right)\\ n_{H_2SO_4}=0,05.0,15=0,0075\left(mol\right)\\ 2NaOH+H_2SO_4\rightarrow Na_2SO_4+2H_2O\\ Vì:\dfrac{0,09}{2}>\dfrac{0,0075}{2}\Rightarrow NaOHdư\\ n_{NaOH\left(dư\right)}=0,09-2.0,0075=0,075\left(mol\right)\\ \left[OH^-\right]=\left[NaOH\right]=\dfrac{0,075}{0,15+0,05}=0,375\left(M\right)\\ \Rightarrow C\)