Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

Phương trình hóa học CaCO3 → CaO + CO2.
a) nCaO = 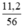 = 0,2 mol.
= 0,2 mol.
Theo PTHH thì nCaCO3 = nCaO = 0,2 (mol)
b) nCaO = 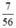 = 0,125 (mol)
= 0,125 (mol)
Theo PTHH thì nCaCO3 = nCaO = 0,125 (mol)
mCaCO3 = M.n = 100.0,125 = 12,5 (g)
c) Theo PTHH thì nCO2 = nCaCO3 = 3,5 (mol)
VCO2 = 22,4.n = 22,4.3,5 = 78,4 (lít)
d) nCO2 = 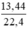 = 0,6 (mol)
= 0,6 (mol)
Theo PTHH nCaO = nCaCO3 = nCO2 = 0,6 (mol)
mCaCO3 = n.M = 0,6.100 = 60 (g)
mCaO = n.M = 0,6.56 = 33,6 (g)

a) \(n_{CaO}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
Ta có: \(n_{CaCO_3}=n_{CaO}=0,2\left(mol\right)\)
b) \(n_{CaO}=\dfrac{7}{56}=0,125\left(mol\right)\)
Ta có : \(n_{CaCO_3}=n_{CaO}=0,125\left(mol\right)\)
=> \(m_{CaCO_3}=0,125.100=12,5\left(g\right)\)
c) \(n_{CO_2}=n_{CaCO_3}=3,5\left(mol\right)\)
=> \(V_{CO_2}=3,5.22,4=78,4\left(lít\right)\)
d) \(n_{CO_2}=\dfrac{13,44}{22,4}=0,6\left(mol\right)\)
Ta có: \(n_{CO_2}=n_{CaCO_3}=n_{CaO}=0,6\left(mol\right)\)
=> \(m_{CaCO_3}=0,6.100=60\left(g\right)\)
\(m_{CaO}=0,6.56=33,6\left(g\right)\)

\(a.\)
\(m_{CaCO_3}=150\cdot80\%=120\left(g\right)\)
\(n_{CaCO_3}=\dfrac{120}{100}=1.2\left(mol\right)\)
\(CaCO_3\underrightarrow{^{^{t^0}}}CaO+CO_2\)
\(1.2...........1.2\)
\(m_{CaO=}=1.2\cdot56=67.2\left(g\right)\)
\(b.\)
\(n_{CO_2}=\dfrac{27.6}{24}=1.15\left(mol\right)\)
\(n_{CaCO_3}=1.15\left(mol\right)\)
\(m_{CaCO_3}=1.15\cdot100=115\left(g\right)\)
\(m_{TC}=115\cdot20\%=23\left(g\right)\)
a, - Khối lượng CaCO3 trong 150g đá là : 120g
=> \(n_{CaCO3}=\dfrac{m}{M}=1,2\left(mol\right)\)
\(PTHH:CaCO_3\rightarrow CaO+CO_2\)
Theo PTHH : \(n_{CaO}=1,2\left(mol\right)\)
\(\Rightarrow m_{vs}=m_{CaO}=n.M=67,2\left(g\right)\)
b, \(n_{CO2}=\dfrac{V}{24}=1,15\left(mol\right)\)
Theo PTHH : \(n_{CaCO3}=1,15\left(mol\right)\)
\(\Rightarrow m_{CaCO3}=n.M=115\left(g\right)\)
=> %Tạp chất là : \(\left(1-\dfrac{115}{150}\right).100\%=\dfrac{70}{3}\%\)
Vậy ...

a) \(n_{O_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
PTHH: 2KClO3 --to--> 2KCl + 3O2
0,2<-------------------0,3
=> \(m_{KClO_3}=0,2.122,5=24,5\left(g\right)\)
b) \(n_{KClO_3}=\dfrac{490}{122,5}=4\left(mol\right)\)
PTHH: 2KClO3 --to--> 2KCl + 3O2
4-------------->4---->6
=> \(m_{KCl}=4.74,5=298\left(g\right)\)
=> \(m_{O_2}=6.32=192\left(g\right)\)
2KClO3 \(\underrightarrow{t^o}\) 2KCl + 3O2
a, \(n_{O_2}=\dfrac{6,72}{22,4}=0,3mol\\ n_{KClO_3}=\dfrac{0,3.2}{3}=0,2mol\\ m_{KClO_3}=0,2.122,5=24,5g\)
b, \(n_{KClO_3}=\dfrac{490}{122,5}=4mol\)
\(\Rightarrow m_{KCl}=4.74,5=298g\)
\(n_{O_2}=\dfrac{4.3}{2}=6mol\\ m_{O_2}=6.32=192g\)

Bài 3 :
$n_{CaO} = \dfrac{168}{56} = 3(kmol)$
$CaCO_3 \xrightarrow{t^o} CaO + CO_2$
$n_{CaCO_3\ pư} = n_{CaO} = 3(kmol)$
$n_{CaCO_3\ đã\ dùng} = \dfrac{3}{80\%} = 3,75(kmol)$
$m_{CaCO_3} = 3,75.100 = 375(kg)$
$m = \dfrac{375}{80\%} = 468,75(kg)$
Bài 2 :
\(m_{CaCO_3}=280\cdot75\%=210\left(kg\right)\)
\(n_{CaCO_3\left(pư\right)}=\dfrac{210}{100}\cdot80\%=1.68\left(kmol\right)\)
\(CaCO_3\underrightarrow{^{^{t^0}}}CaO+CO_2\)
\(1.68.........1.68......1.68\)
\(m_{CaO}=1.68\cdot56=94.08\left(kg\right)\)
\(V_{CO_2}=1.68\cdot22.4=37.632\left(l\right)=0.037632\left(m^3\right)\)

1. Na + 1/2O2 -> NaO
Al2O3 + 6HCl -> 2AlCl3 + 3H2O
AgNO3 + NaCl -> AgCl + NaNO3
CuSO4 + 2NaOH -> Na2SO4 + Cu(OH)2

a. m2ycao là gì vậy bạn
\(n_{CaO}=\dfrac{7}{56}=0,125mol\\ CaCO_3\xrightarrow[]{t^0}CaO+CO_2\\ n_{CaCO_3}=n_{CaO}=0,125mol\\ m_{CaCO_3}=0,125.100=12,5g\\ c.n_{CO_2}=\dfrac{2,479}{24,79}=0,1mol\\ n_{CaCO_3}=n_{CaO}=n_{CO_2}=0,1mol\\ m_{CaCO_3}=0,1.100=10g\\ m_{CaO}=0,1.56=5,6g\)
 2KCl+3O2
2KCl+3O2