
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.



\(n_{Al}=\dfrac{10,8}{27}=0,4\left(mol\right)\)
PTHH :
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\uparrow\)
0,4 1,2 0,4 0,6
\(a,V_{H_2}=0,6.22,4=13,44\left(l\right)\)
\(b,m_{HCl}=1,2.36,5=43,8\left(g\right)\)
\(c,m_{AlCl_3}=133,5.0,4=53,4\left(g\right)\)
\(m_{ddHCl}=\dfrac{43,8.100}{10}=438\left(g\right)\)
\(m_{ddAlCl_3}=10,8+438-\left(0,6.2\right)=447,6\left(g\right)\)
\(C\%=\dfrac{53,8}{447,6}.100\%\approx12,02\%\)

\(2A+2nH_2SO_4\underrightarrow{to}A_2\left(SO_4\right)_n+nSO_2+2nH_2O\)

\(M_{SO_{2}}\)=32+16.2=64g/mol
\(M_{O_{2}}\)=16.2=32 g/mol
\(d_{M_{SO_{2}}/{M_{O_{2}}}}\)= \(\dfrac{M_{SO_{2}}}{M_{O_{2}}}\)=\(\dfrac{64}{32}\)=2
Vậy SO2 nặng hơn O2 2 lần


\(1.4P+5O_2\overset{t^o}{--->}2P_2O_5\left(4:5:2\right)\)
\(2.2Zn+O_2\overset{t^o}{--->}2ZnO\left(2:1:2\right)\)
\(3.2Fe+3Cl_2\overset{t^o}{--->}2FeCl_3\left(2:3:2\right)\)
\(4.N_2+3H_2\overset{t^o}{--->}2NH_3\left(1:3:2\right)\)
\(5.2Al+3H_2SO_4--->Al_2\left(SO_4\right)_3+3H_2\left(2:3:1:3\right)\)
\(6.P_2O_5+3H_2O--->2H_3PO_4\left(1:3:2\right)\)
\(7.Na_2O+H_2O--->2NaOH\left(1:1:2\right)\)
\(8.Fe_2O_3+6HCl--->2FeCl_3+3H_2O\left(1:6:2:3\right)\)
\(9.2Al\left(OH\right)_3+3H_2SO_4--->Al_2\left(SO_4\right)_3+6H_2O\left(2:3:1:6\right)\)
\(10.MgCl_2+2AgNO_3--->Mg\left(NO_3\right)_2+2AgCl\downarrow\left(1:2:1:2\right)\)
\(11.2FeCl_3+3Ba\left(OH\right)_2--->2Fe\left(OH\right)_3\downarrow+3BaCl_2\left(2:3:2:3\right)\)
\(12.2Al+3CuCl_2--->2AlCl_3+3Cu\downarrow\left(2:3:2:3\right)\)
\(13.2Al+Fe_2O_3\overset{t^o}{--->}2Fe+Al_2O_3\left(2:1:2:1\right)\)
\(14.2Fe\left(OH\right)_3\overset{t^o}{--->}Fe_2O_3+3H_2O\left(2:1:3\right)\)
\(15.2KClO_3\xrightarrow[MnO_2]{t^o}2KCl+3O_2\left(2:2:3\right)\)
\(16.CH_4+2O_2\overset{t^o}{--->}CO_2+2H_2O\left(1:2:1:2\right)\)
\(17.4K+O_2\overset{t^o}{--->}2K_2O\left(4:1:2\right)\)
\(18.2Al+3Cl_2\overset{t^o}{--->}2AlCl_3\left(2:3:2\right)\)
\(19.Mg+2HCl--->MgCl_2+H_2\left(1:2:1:1\right)\)
\(20.3H_2+Fe_2O_3\overset{t^o}{--->}2Fe+3H_2O\left(3:1:2:3\right)\)
\(21.4Na+O_2\overset{t^o}{--->}2Na_2O\left(4:1:2\right)\)
\(22.2Mg+O_2\overset{t^o}{--->}2MgO\left(2:1:2\right)\)
\(23.2Al+6HCl--->2AlCl_3+3H_2\left(2:6:2:3\right)\)
\(24.CuO+2HCl--->CuCl_2+H_2O\left(1:2:1:1\right)\)
\(25.Mg+2AgNO_3--->Mg\left(NO_3\right)_2+2Ag\downarrow\left(1:2:1:2\right)\)
\(26.FeCl_3+3KOH--->3KCl+Fe\left(OH\right)_3\downarrow\left(1:3:3:1\right)\)

Sai:
CaOH -> Ca (OH)2
Na2PO4 -> Na3PO4
H3S -> H2S
NaO -> Na2O
H2NO2 -> HNO2
Còn lại là đúng rồi bạn




Có mấy cthh sai như Al2S, Mg3(PO3)2 thì mình không xếp vào ha
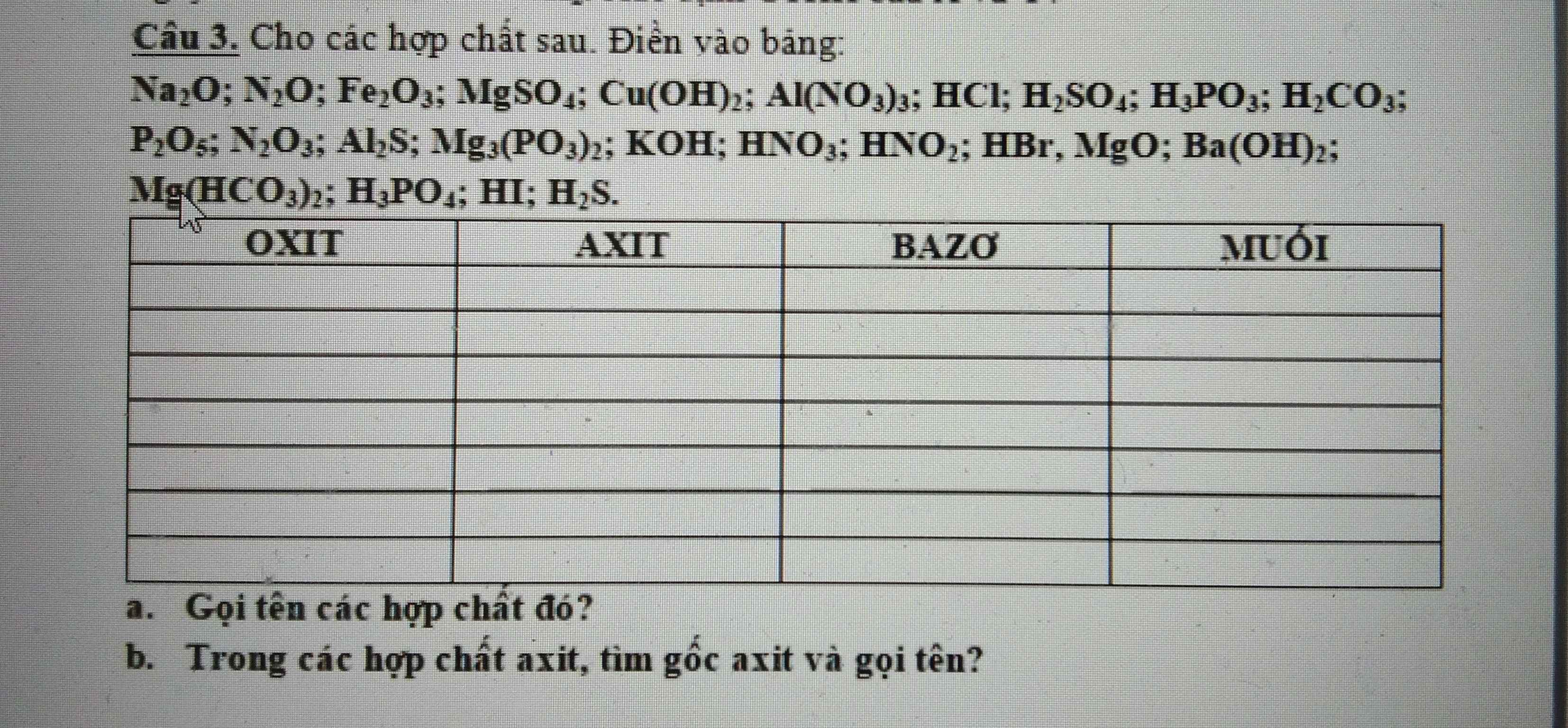
Câu 3:
b)
Oxit:
Na2O: natri oxit
N2O: đinitơ oxit
Fe2O3: sắt (III) oxit
P2O5: điphotpho pentaoxit
N2O3: đinitơ trioxit
MgO: magie oxit
Axit:
HCl: axit clohiđric
H2SO4: axit sunfuric
H3PO3: axit photphorơ
H2CO3: axit cacbonic
HNO3: axit nitrat
HNO3: axit nitric
HBr: axit bromhiđric
H3PO4: axit photphorơ
HI: axit iothiđric
H2S: axit hiđro sunfua
Bazơ:
Cu(OH)2: đồng (II) hiđroxit
KOH: Kali hiđroxit
Ba(OH)2: bari hiđroxit
Muối:
MgSO4: magie sunfat
Al(NO3)3: nhôm nitrat
Al2S: nhôm sunfua
Mg3(PO3)2: magie photphit
Mg(HCO3)2: magie hiđrocacbonat
Gốc axit:
NO3, SO4, I, Cl, Br, S, HCO3, CO3, PO3, PO4