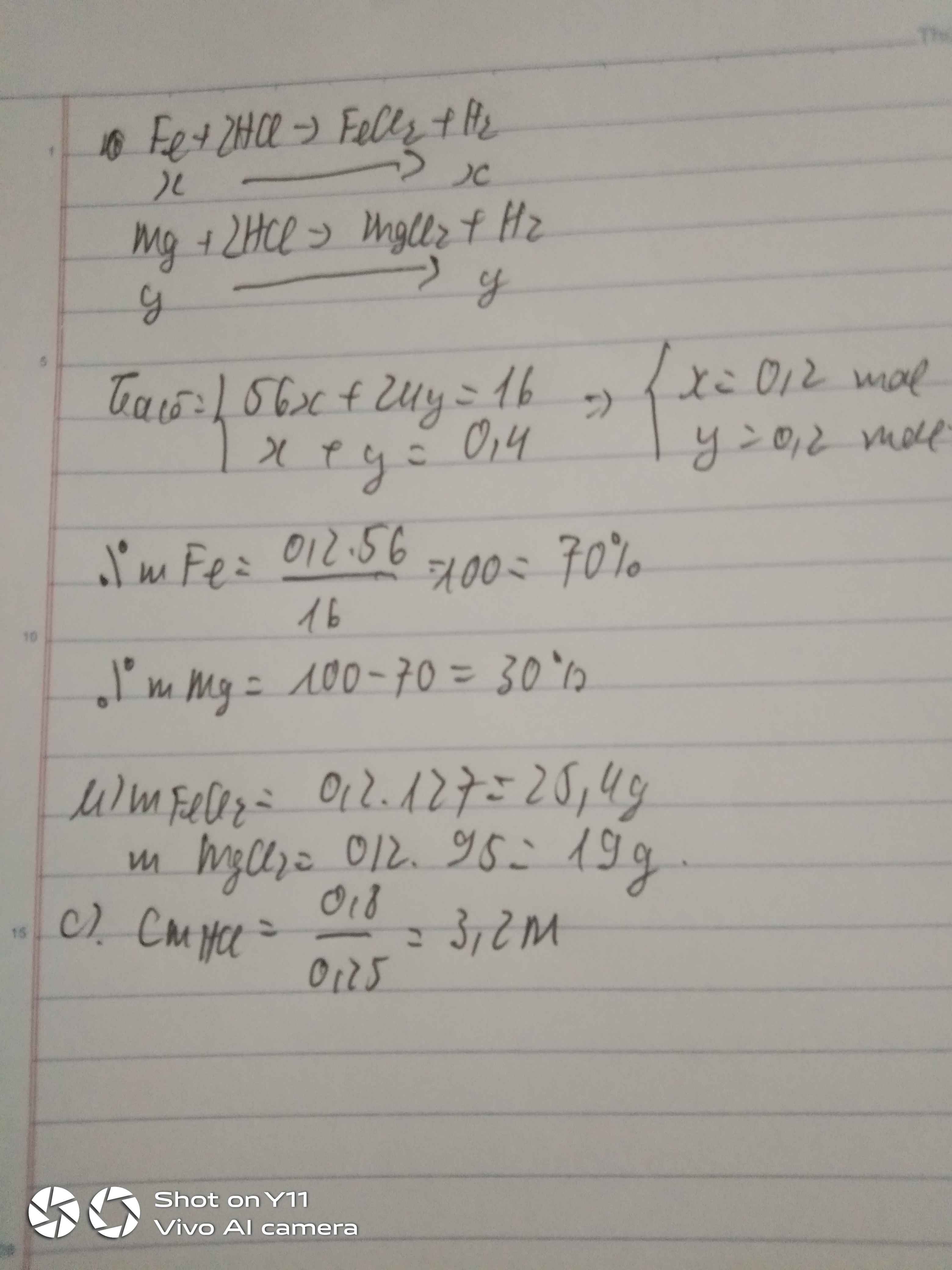Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a)
| $Fe^0 \to Fe^{+3} + 3e$ | x2 |
| $Cl_2 + 2e \to 2Cl^-$ | x3 |
$2Fe + 3Cl_2 \xrightarrow{t^o} 2FeCl_3$
b)
$n_{Fe} = \dfrac{28}{56} = 0,5(mol)$
$n_{Cl_2} = \dfrac{2,479}{24,79} = 0,1(mol)$
Ta thấy :
$n_{Fe} :2>n_{Cl_2}:3$ nên $Fe$ dư
$n_{FeCl_3} = \dfrac{2}{3}n_{Cl_2} = \dfrac{0,2}{3}(mol)$
$\Rightarrow m_{FeCl_3} = \dfrac{0,2}{3}.162,5 = 10,83(gam)$

\(1.a)QToxh:S^{+4}\rightarrow S^{+6}+2e|\times3\\ QTkhử:N^{+5}+3e\rightarrow N^{+2}|\times2\\ 3SO_2+2HNO_3+2H_2O\rightarrow2NO+3H_2SO_4\\ b)QToxh:\overset{0}{Fe}\rightarrow Fe^{3+}+3e|\times1\\ QTkhử:N^{+5}+1e\rightarrow N^{+4}|\times3\\ Fe+6HNO_3\rightarrow Fe\left(NO_3\right)_3+3NO_2+3H_2O\)

\(n_{H_2}=\dfrac{7,84}{22,4}=0,35mol\)
Gọi \(\left\{{}\begin{matrix}n_{Fe}=x\\n_{Zn}=y\end{matrix}\right.\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
x x ( mol )
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
y y ( mol )
Ta có:
\(\left\{{}\begin{matrix}56x+65y=21,4\\x+y=0,35\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}x=0,15\\y=0,2\end{matrix}\right.\)
\(\Rightarrow m_{Fe}=0,15.56=8,4g\)
\(\Rightarrow m_{Zn}=0,2.65=13g\)
\(\%m_{Fe}=\dfrac{8,4}{21,4}.100=39,25\%\)
\(\%m_{Zn}=100\%-39,25\%=60,75\%\)
\(m_{FeCl_2}=0,15.127=19,05g\)
\(m_{ZnCl_2}=0,2.136=27,2g\)

Gọi số mol Zn, Fe là a, b (mol)
=> 65a + 56b = 23,75 (1)
\(n_{H_2}=\dfrac{8,96}{22,4}=0,4\left(mol\right)\)
PTHH: Zn + 2HCl --> ZnCl2 + H2
a--------------->a------->a
Fe + 2HCl --> FeCl2 + H2
b----------------->b----->b
=> a + b = 0,4 (2)
(1)(2) => a = 0,15 (mol); b = 0,25 (mol)
=> mZn = 0,15.65 = 9,75 (g); mFe = 0,25.56 = 14 (g)
\(\left\{{}\begin{matrix}\%m_{Zn}=\dfrac{9,75}{23,75}.100\%=41,05\%\\\%m_{Fe}=\dfrac{14}{23,75}.100\%=58,95\%\end{matrix}\right.\)
b) mZnCl2 = 0,15.136 = 20,4 (g)
mFeCl2 = 0,25.127 = 31,75 (g)
=> mmuối = 20,4 + 31,75 = 52,15 (g)

Câu 2:
\(n_{AgNO_3}=\dfrac{25,5}{170}=0,15(mol)\\ PTHH:NaCl+AgNO_3\to AgCl\downarrow +NaNO_3\\ \Rightarrow m_{NaCl}=0,15.58,5=8,775(g)\)
Câu 3:
\(a,\)Đặt \(\begin{cases} n_{Mg}=x(mol)\\ n_{Zn}=y(mol) \end{cases} \Rightarrow 24x+65y=15,75(1)\)
\(PTHH:Mg+2HCl\to MgCl_2+H_2\\ Zn+2HCl\to ZnCl_2+H_2\\ \Rightarrow 95x+136y=44,15(2)\\ (1)(2)\Rightarrow \begin{cases} x=0,25(mol)\\ y=0,15(mol) \end{cases} \Rightarrow \begin{cases} \%_{Mg}=\dfrac{0,25.24}{15,75}.100\%=38,1\%\\ \%_{Zn}=100\%=38,1\%=61,9\% \end{cases} \)
\(b,\Sigma n_{HCl}=2n_{Mg}+2n_{Zn}=0,8(mol)\\ \Rightarrow m_{dd_{HCl}}=\dfrac{36,5.0,8}{10\%}=292(g)\)

$PTHH:2KMnO_4+16HCl\to 2KCl+2MnCl_2+5Cl_2\uparrow+8H_2O(1)$
$2Fe+3Cl_2\xrightarrow{t^o}2FeCl_3(2)$
$n_{KMnO_4}=\dfrac{47,4}{158}=0,3(mol);n_{Fe}=\dfrac{16,8}{56}=0,3(mol)$
Theo PT: $n_{Cl_2(1)}=0,75(mol)\Rightarrow n_{Cl_2(2)}=0,75(mol)$
Lập tỉ lệ: $\dfrac{n_{Cl_2(2)}}{3}>\dfrac{n_{Fe}}{2}\Rightarrow Cl_2$ dư
$\Rightarrow n_{FeCl_3}=n_{Fe}=0,3(mol)$
$\Rightarrow m_{FeCl_3}=0,3.162,5=48,75(g)$