Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{Br_2}=\dfrac{4}{160}=0,025\left(mol\right);n_{hh}=\dfrac{2,8}{22,4}=0,125\left(mol\right)\)
PTHH: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
0,025<-0,125
\(\Rightarrow\left\{{}\begin{matrix}\%V_{C_2H_4}=\dfrac{0,025}{0,125}.100\%=20\%\\\%V_{CH_4}=100\%-20\%=80\%\end{matrix}\right.\)

a, PT: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
Khí thoát ra khỏi bình là CH4 (metan).
b, Ta có: \(\left\{{}\begin{matrix}\%V_{CH_4}=\dfrac{1,12}{5,6}.100\%=20\%\\\%V_{C_2H_4}=100-20=80\%\end{matrix}\right.\)
c, Ta có: \(V_{C_2H_4}=5,6.80\%=4,48\left(l\right)\)
\(\Rightarrow n_{C_2H_4}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
Theo PT: \(n_{Br_2}=n_{C_2H_4}=0,2\left(mol\right)\)
\(\Rightarrow m_{Br_2}=0,2.160=32\left(g\right)\)
Bạn tham khảo nhé!

1. \(n_{Br_2}=0,4.0,5=0,2\left(mol\right)\)
PT: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
Theo PT: \(n_{C_2H_4}=n_{Br_2}=0,2\left(mol\right)\Rightarrow V_{C_2H_4}=0,2.22,4=4,48\left(l\right)\)
2. \(n_{C_2H_4Br_2}=\dfrac{9,4}{188}=0,05\left(mol\right)\)
PT: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
Theo PT: \(n_{C_2H_4}=n_{C_2H_4Br_2}=0,05\left(mol\right)\)
\(\Rightarrow\%V_{C_2H_4}=\dfrac{0,05.22,4}{1,4}.100\%=80\%\)
\(\Rightarrow\%V_{CH_4}=100-80=20\%\)

a, \(C_2H_2+2Br_2\rightarrow C_2H_2Br_4\)
Ta có: \(n_{Br_2}=\dfrac{64}{160}=0,4\left(mol\right)\)
Theo PT: \(n_{C_2H_2}=\dfrac{1}{2}n_{Br_2}=0,2\left(mol\right)\)
Mà: \(n_X=\dfrac{10,08}{22,4}=0,45\left(mol\right)\Rightarrow n_{CH_4}=0,25\left(mol\right)\)
\(\Rightarrow V_{CH_4}=0,25.22,4=5,6\left(l\right)\)
c, \(CH_4+2O_2\underrightarrow{t^o}CO_2+2H_2O\)
\(2C_2H_2+5O_2\underrightarrow{t^o}4CO_2+2H_2O\)
Theo PT: \(n_{CO_2}=n_{CH_4}+2n_{C_2H_2}=0,65\left(mol\right)\)
PT: \(CO_2+Ca\left(OH\right)_2\rightarrow CaCO_{3\downarrow}+H_2O\)
Theo PT: \(n_{CaCO_3}=n_{CO_2}=0,65\left(mol\right)\Rightarrow m_{CaCO_3}=0,65.100=65\left(g\right)\)

a, - Khí pư với Brom là C2H4
PT: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
b, Ta có: \(n_{Br_2}=\dfrac{16}{160}=0,1\left(mol\right)\)
Theo PT: \(n_{C_2H_4}=n_{Br_2}=0,1\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{C_2H_4}=\dfrac{0,1.28}{4}.100\%=70\%\\\%m_{CH_4}=100-70=30\%\end{matrix}\right.\)

nhh = 6.72/22.4 = 0.3 (mol)
nBr2 = 64/160 = 0.4 (mol)
nC2H4 = a (mol) . nC2H2 = b (mol)
C2H2 + 2Br2 => C2H2Br4
C2H4 + Br2 => C2H4Br2
=> a + b = 0.3
a + 2b = 0.4
=> a =0.2 , b = 0.1
%VC2H4 = 0.2/0.3 * 100% = 66.67%
%VC2H2 = 33.33%
a, vì CH4 là hidrocacbon no => không xảy ra phản ứng với Brom
pt: C2H4 + Br2 -> C2H4Br2
1 1 1
nBr2 = m/M = 6,4/160 = 0,04 mol => nC2H4 = 0,04 mol
=> VC2H4 = n x 22,4 = 0,04 x 22,4 = 0,896 lit
=> VCH4 = Vhh - VC2H4 = 6,72 - 0,896 = 5,824 lit
b, C%VC2H4 = VC2H4/Vhh = 0,896/6,72 X 100 = 13,33%
=> C%VCH4 = Vhh - VC2H4 = 100% - 13,33% = 86,67%

a) \(C_2H_4 + Br_2 \to C_2H_4Br_2\)
b)
\(n_{C_2H_4} = n_{Br_2} = \dfrac{5,6}{160} = 0,035(mol)\\ \%V_{C_2H_4} = \dfrac{0,035.22,4}{0,86}.100\% = 91,16\%\\ \%V_{CH_4} = 100\% - 91,16\% = 8,84\%\)
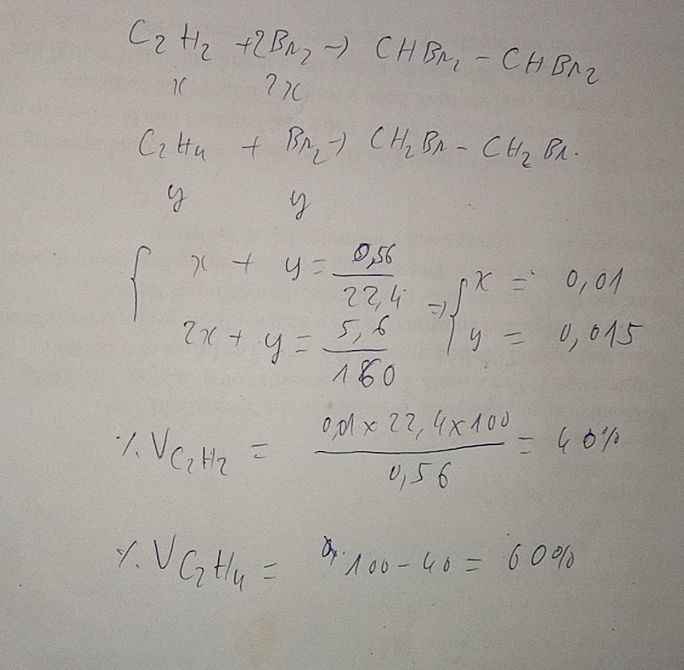
a) \(n_{Br_2\left(p\text{ư}\right)}=\dfrac{200.8\%}{160}=0,1\left(mol\right);n_{hh}=\dfrac{7,437}{24,79}=0,3\left(mol\right)\)
PTHH: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
0,1<-----0,1
b) \(\left\{{}\begin{matrix}\%V_{C_2H_4}=\dfrac{0,1}{0,3}.100\%=33,33\%\\\%V_{CH_4}=100\%-33,33\%=66,67\%\end{matrix}\right.\)
c) \(n_{CH_4}=0,3-0,1=0,2\left(mol\right)\)
PTHH: \(CH_4+2O_2\xrightarrow[]{t^o}CO_2+2H_2O\)
0,2--->0,4
\(C_2H_4+3O_2\xrightarrow[]{t^o}2CO_2+2H_2O\)
0,1--->0,3
\(\Rightarrow V_{O_2}=\left(0,3+0,4\right).24,79=17,353\left(l\right)\)