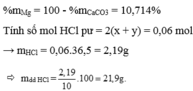Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(Mg+2HCl\rightarrow MgCl_2+H_2\)
\(MgCO_3+2HCl\rightarrow MgCl_2+H_2O+CO_2\)
a, \(n_A=0,5\left(mol\right)=n_{H_2}+n_{CO_2}\)
\(m_{hh}=33=24n_{Mg}+84n_{MgCO_3}=84n_{CO_2}+24n_{H_2}\)
\(\Rightarrow\left\{{}\begin{matrix}n_{H_2}=0,15\left(30\%\right)\\n_{CO_2}=0,35\left(70\%\right)\end{matrix}\right.\) ( mol )
b, Có \(n_{HCl}=2n_{H_2}+2n_{CO_2}=1\left(mol\right)\)
Mà \(n_{HClbd}=1,6\left(mol\right)\)
\(\Rightarrow n_{HCldu}=0,6\left(mol\right)\)
Lại có : \(m_{ddsau}=m_{hh}+m_{ddHCl}-m_{hhkhi}=33+880-0,15.2-0,35.44=897,3\left(g\right)\)
\(\Rightarrow\left\{{}\begin{matrix}C\%_{MgCl_2}=\dfrac{0,5.\left(24+71\right)}{897,3}.100\%\approx5,3\%\\C\%_{HCldu}=\dfrac{0,6\left(35,5+1\right)}{897,3}.100\%\approx2,44\%\end{matrix}\right.\)

nH2 = 6,72/22,4 = 0,3 (mol)
PTHH: 2Al + 6HCl -> 2AlCl3 + 3H2
nAl = 0,3 : 3 . 2 = 0,2 (mol)
nHCl (Al) = 0,3 . 2 = 0,6 (mol)
mAl = 0,2 . 27 = 5,4 (g)
%mAl = 5,4/25,65 = 20,05%
%mZnO = 100% - 20,05% = 79,95%
mZnO = 25,65 - 5,4 = 20,25 (g)
nZnO = 20,25/81 = 0,25 (mol)
PTHH: ZnO + 2HCl -> ZnCl2 + H2O
nHCl (ZnO) = 0,25 . 2 = 0,5 (mol)
nHCl (đã dùng) = 0,6 + 0,5 = 1,1 (mol)
CMddHCl = 1,1/0,1008 = 10,9M
C% = (10,9 . 36,5)/(10 . 1,19) = 33,43%

PTHH: \(Zn+2HCl\rightarrow ZnCl_2+H_2\uparrow\)
\(Al_2O_3+6HCl\rightarrow2AlCl_3+3H_2O\)
a) Ta có: \(n_{H_2}=\dfrac{8,96}{22,4}=0,4\left(mol\right)=n_{Zn}\)
\(\Rightarrow\%m_{Zn}=\dfrac{0,4\cdot65}{36,2}\cdot100\%\approx71,23\%\) \(\Rightarrow\%m_{Al_2O_3}=28,77\%\)
c) Ta có: \(n_{Al_2O_3}=\dfrac{36,2-0,4\cdot65}{102}=0,1\left(mol\right)\)
Theo PTHH: \(n_{HCl}=2n_{Zn}+6n_{Al_2O_3}=1,4\left(mol\right)\)
\(\Rightarrow m_{ddHCl}=\dfrac{1,4\cdot36,5}{10\%}=511\left(g\right)\) \(\Rightarrow V_{ddHCl}=\dfrac{511}{1,1}\approx464,5\left(ml\right)=0,4645\left(l\right)\)
c) Theo PTHH: \(\left\{{}\begin{matrix}n_{ZnCl_2}=0,4\left(mol\right)\\n_{AlCl_3}=0,2\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}C_{M_{ZnCl_2}}=\dfrac{0,4}{0,4645}\approx0,86\left(M\right)\\C_{M_{AlCl_3}}=\dfrac{0,2}{0,4645}\approx0,43\left(M\right)\end{matrix}\right.\)

$a)PTHH:2Al+6HCl\to 2AlCl_3+3H_2$
$n_{H_2}=\dfrac{5,04}{22,4}=0,225(mol)$
$\Rightarrow n_{Al}=0,15(mol)$
$\Rightarrow \%m_{Al}=\dfrac{0,15.27}{9,45}.100\%\approx 42,86\%$
$\Rightarrow \%m_{Cu}=100-42,86=57,14\%$
$b)$ Theo PT: $n_{HCl}=2n_{H_2}=0,45(mol)$
$\Rightarrow C_{M_{HCl}}=\dfrac{0,45.110\%}{0,5}=0,99M$

1)
a)
$CaO + 2HCl \to CaCl_2 + H_2O$
$CaCO_3 + 2HCl \to CaCl_2 + CO_2 + H_2O$
$n_{CaCO_3} = n_{CO_2} = 0,2(mol)$
$n_{CaCl_2} = 0,3(mol)$
Suy ra:
$n_{CaO} = 0,3 - 0,2 = 0,1(mol)$
$\%m_{CaO} = \dfrac{0,1.56}{0,1.56 + 0,2.100}.100\% = 21,875\%$
$\%m_{CaCO_3} = 78,125\%$
b)
$m_{dd} = 0,1.56 + 0,2.100 + 50 - 0,2.44 = 66,8(gam)$
$C\%_{CaCl_2} = \dfrac{33,3}{66,8}.100\% = 49,85\%$
Câu 4 :
a)
Gọi $n_{Fe} = a(mol) ; n_{MgO} = b(mol)$
Suy ra: $56a + 40b = 19,2(1)$
$Fe + 2HCl \to FeCl_2 + H_2$
$MgO + 2HCl \to MgCl_2 + H_2O$
Theo PTHH : $n_{HCl} = 2a + 2b = 0,4.2 = 0,8(2)$
Từ (1)(2) suy ra a = b = 0,2
$\%m_{Fe} = \dfrac{0,2.56}{19,2}.100\% = 58,33\%$
$\%m_{MgO} = 100\% -58,33\% = 41,67\%$
b)
$n_{FeCl_2} = a = 0,2(mol)$
$n_{MgCl_2} = b = 0,2(mol)$
$m_{muối} = 0,2.127 + 0,2.95 = 44,4(gam)$

Ta có: \(\left\{{}\begin{matrix}n_{CO_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\\n_{CaCl_2}=\dfrac{33,3}{111}=0,3\left(mol\right)\end{matrix}\right.\)
PTHH: \(CaCO_3+2HCl\rightarrow CaCl_2+CO_2\uparrow+H_2O\)
0,2_____0,4_____0,2____0,2_____0,2 (mol)
\(CaO+2HCl\rightarrow CaCl_2+H_2O\)
0,1_____0,2_____0,1____0,1 (mol)
Ta có: \(\left\{{}\begin{matrix}m_{CaO}=0,1\cdot56=5,6\left(g\right)\\m_{CaCO_3}=0,2\cdot100=20\left(g\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}\%m_{CaO}=\dfrac{5,6}{5,6+20}\cdot100\%=21,875\%\\\%m_{CaCO_3}=78,125\%\end{matrix}\right.\)
Mặt khác: \(m_{CO_2}=0,2\cdot44=8,8\left(g\right)\)
\(\Rightarrow m_{dd\left(sau.p/ứ\right)}=m_{CaO}+m_{CaCO_3}+m_{ddHCl}-m_{CO_2}=66,8\left(g\right)\)
\(\Rightarrow C\%_{CaCl_2}=\dfrac{33,3}{66,8}\cdot100\%\approx49,85\%\)
nCO2=\(\dfrac{4,48}{22,4}=0,2\) mol
nCaCl2=\(\dfrac{33,3}{111}=0,3\)
CaCO2 + 2HCl → CaCl2 + CO2 + H2O
0,2 ← 0,2 ← 0,2
CaO + 2HCl → CaCl2 + H2O
0,1 ← 0,1
a) % CaO=\(\dfrac{0,1.56}{0,1.56+0,2.100}.100\%=21,875\%\)
% CaCO3 =100% - 21,875%= 78,125%
b) a = mCaO+mCaCO3 =0,1.56+0,2.100=25,6g
mdd sau pư= a + mddHCl - mCO2
= 25,6 + 50 - 0,2.44=66,8g
C%CaCl2=\(\dfrac{33,3}{66,8}.100\%\simeq49,85\%\)

a) Gọi số mol Zn, Fe là a, b (mol)
=> 65a + 56b = 7,35 (1)
PTHH: Zn + 2HCl --> ZnCl2 + H2
a---->2a------->a------>a
Fe + 2HCl --> FeCl2 + H2
b------>2b----->b------>b
=> \(a+b=\dfrac{2,688}{22,4}=0,12\left(mol\right)\)
=> a + b = 0,12 (2)
(1)(2) => a = 0,07; b = 0,05
=> \(\left\{{}\begin{matrix}\%m_{Zn}=\dfrac{0,07.65}{7,35}.100\%=61,9\%\\\%m_{Fe}=\dfrac{0,05.56}{7,35}.100\%=38,1\%\end{matrix}\right.\)
b) nHCl(dư) = 0,3.1 - 0,07.2 - 0,05.2 = 0,06 (mol)
PTHH: Ca(OH)2 + 2HCl --> CaCl2 + 2H2O
0,03<-----0,06
=> \(x=C_{M\left(ddCa\left(OH\right)_2\right)}=\dfrac{0,03}{0,1}=0,3M\)
c) Chất rắn thu được là Fe2O3
Bảo toàn Fe: \(n_{Fe_2O_3}=0,025\left(mol\right)\)
=> \(a=m_{Fe_2O_3}=0,025.160=4\left(g\right)\)
Kết tủa thu được là Fe(OH)2
Bảo toàn Fe: \(n_{Fe\left(OH\right)_2}=0,05\left(mol\right)\)
=> \(m=m_{Fe\left(OH\right)_2}=0,05.90=4,5\left(g\right)\)