Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) $n_{CaCO_3} = 0,15(mol)$
$CaCO_3 + 2HCl \to CaCl_2 + CO_2 + H_2O$
$n_{HCl} = 2n_{CaCO_3} = 0,3(mol)$
$m_{dd\ HCl} = \dfrac{0,3.36,5}{7,3\%} = 150(gam)$
b)
$n_{CaCl_2} = n_{CO_2} = n_{CaCO_3} =0,15(mol)$
$V_{CO_2} = 0,15.22,4 = 3,36(lít)$
c)
$m_{dd} = 15 + 150 - 0,15.44 = 158,4(gam)$
$C\%_{CaCl_2} = \dfrac{0,15.111}{158,4}.100\% = 10,51\%$

Bài 5
Fe + 2CH3COOH \(\rightarrow\) (CH3COO)2Fe + H2(1)
nCH3COOH = \(\dfrac{4,5}{60}=0,075mol\)
a) THeo pt: n(CH3COO)2Fe = \(\dfrac{1}{2}.nCH_3COOH=0,0375mol\)
=> m = 6,525g
c) Theo pt (1) nH2 = 1/2nCH3COOH = 0,0375 mol
2H2 + O2 \(\xrightarrow[]{t^o}\) 2H2O
Theo pt: nO2 = 0,5nH2 = 0,01875mol
=> VO2 = 0,42 lít
=> Vkk = 0,42.5 = 2,1 lít

\(m_{CH_3COOH}=6\%.200=12\left(g\right)\\ \rightarrow n_{CH_3COOH}=\dfrac{12}{60}=0,2\left(mol\right)\\ n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\)
PTHH: \(2CH_3COOH+Zn\rightarrow\left(CH_3COO\right)_2Zn+H_2\uparrow\)
LTL: \(\dfrac{0,2}{2}>0,2\rightarrow\) Zn dư
Theo pthh: \(n_{\left(CH_3COO\right)_2Zn}=n_{Zn\left(pư\right)}=n_{H_2}=\dfrac{1}{2}n_{CH_3COOH}=\dfrac{1}{2}.0,2=0,1\left(mol\right)\)
\(\rightarrow\left\{{}\begin{matrix}m_{H_2}=0,1.2=0,2\left(g\right)\\m_{Zn\left(pư\right)}=0,1.65=6,5\left(g\right)\\m_{\left(CH_3COO\right)_2Zn}=0,1.183=18,3\left(g\right)\end{matrix}\right.\)
\(\rightarrow m_{dd}=200+6,5-0,2=206,3\left(g\right)\\ \rightarrow C\%_{\left(CH_3COO\right)_2Zn}=\dfrac{18,3}{206,3}=8,87\%\)

\(a,PTHH:Na_2CO_3+2HCl\rightarrow2NaCl+H_2O+CO_2\uparrow\\ b,n_{Na_2CO_3}=\dfrac{15,9}{106}=0,15\left(mol\right)\\ \Rightarrow n_{HCl}=0,3\left(mol\right)\\ \Rightarrow m_{CT_{HCl}}=0,3\cdot36,5=10,95\left(g\right)\\ \Rightarrow C\%_{HCl}=\dfrac{10,95}{200}\cdot100\%=5,475\%\\ c,n_{CO_2}=0,15\left(mol\right)\\ \Rightarrow V_{CO_2\left(đkc\right)}=0,15\cdot24,79=3,7185\left(l\right)\\ d,m_{CO_2}=0,15\cdot44=6,6\left(g\right)\\ n_{NaCl}=0,3\left(mol\right);n_{H_2O}=0,15\left(mol\right)\\ \Rightarrow\left\{{}\begin{matrix}m_{CT_{NaCl}}=0,3\cdot58,5=17,55\left(g\right)\\m_{H_2O}=0,15\cdot18=2,7\left(g\right)\end{matrix}\right.\\ m_{dd_{NaCl}}=15,9+200-2,7-6,6=206,6\left(g\right)\\ \Rightarrow C\%_{NaCl}=\dfrac{17,55}{206,6}\cdot100\%\approx8,49\%\)

Pt: \(2CH_3COOH+Zn\rightarrow\left(CH_3COO\right)_2Zn+H_2\)
\(n_{\left(CH_3COO\right)_2Zn}=\dfrac{14,2}{183}\approx0.077mol\)
Theo pt: nH2 = n(CH3COO)2Zn = 0,077mol
=> VH2 = 1,7248l
b) Theo pt: nCH3COOH = 2n(CH3COO)2Zn = 0,154 mol
=> CMCH3COOH = 0,154 : 0,25 = 0,616M

a) \(n_{Na_2CO_3}=\dfrac{21,2}{106}=0,2\left(mol\right)\)
PTHH: Na2CO3 + 2CH3COOH --> 2CH3COONa + CO2 + H2O
0,2--------->0,4--------------->0,4------->0,2
=> VCO2 = 0,2.22,4 = 4,48 (l)
b) \(m_{dd.CH_3COOH}=\dfrac{0,4.60}{12\%}=200\left(g\right)\)
c) mdd sau pư = 21,2 + 200 - 0,2.44 = 212,4 (g)
=> \(C\%_{muối}=\dfrac{0,4.82}{212,4}.100\%=15,44\%\)

a) \(n_{\left(CH_3COO\right)_2Mg}=\dfrac{7,1}{142}=0,05\left(mol\right)\)
PTHH: 2CH3COOH + Mg --> (CH3COO)2Mg + H2
0,1<----------------------0,05------->0,05
=> VH2 = 0,05.22,4 = 1,12 (l)
b) \(C_{M\left(dd.CH_3COOH\right)}=\dfrac{0,1}{0,025}=4M\)

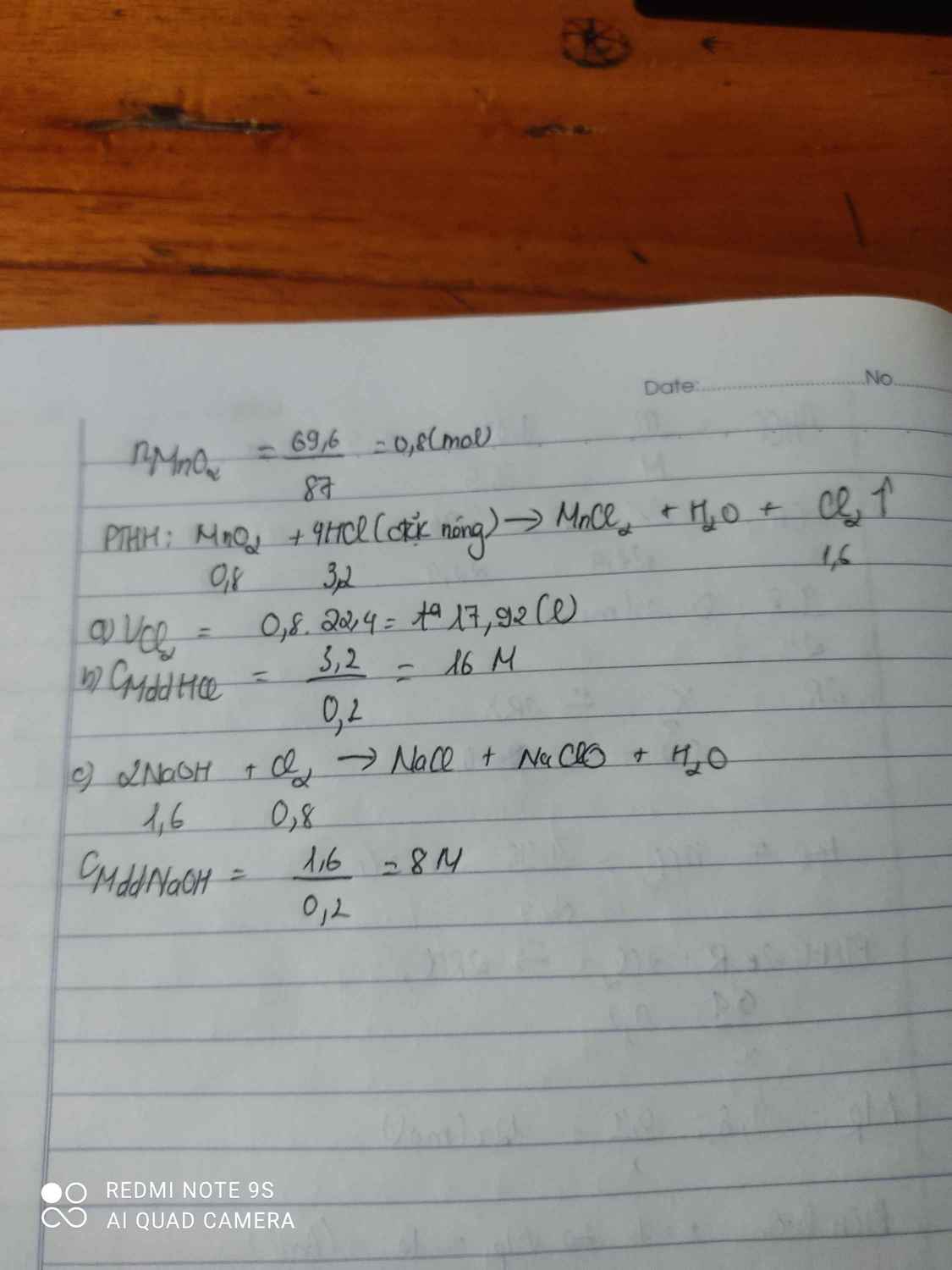
\(n_{CaCO_3}=\dfrac{6}{100}=0,06mol\)
\(n_{CH_3COOH}=\dfrac{200}{60}=3,33mol\)
\(2CH_3COOH+CaCO_3\rightarrow\left(CH_3COO\right)_2Ca+CO_2+H_2O\)
3,33 > 0,06 ( mol )
0,06 0,06 0,06 ( mol )
\(V_{CO_2}=0,06.22,4=1,344l\)
\(m_{\left(CH_3COO\right)_2Ca}=0,06.158=9,48g\)
\(m_{ddspứ}=200+6-0,06.12=205,28g\)
\(C\%_{\left(CH_3COO\right)_2Ca}=\dfrac{9,48}{205,28}.100=4,61\%\)
\(n_{CaCO_3}=\dfrac{6}{100}=0,06\left(mol\right)\\ n_{CH_3C\text{OO}H}=\dfrac{200}{60}=3,3\left(G\right)\\ pthh:CaCO_3+2CH_3C\text{OO}H\rightarrow Ca\left(CH_3C\text{OO}\right)_2+H_2O+CO_2\)
LTL : \(\dfrac{0,06}{1}< \dfrac{3,3}{2}\)
=> CaCO3 hết
theo pthh : \(n_{CO_2}=n_{CaCO_3}=0,06\left(mol\right)\)
\(\Rightarrow V_{CO_2}=0,06.22,4=1,344\left(l\right)\)
\(\Rightarrow C\%=\dfrac{6}{200}.100\%=3\%\dfrac{\dfrac{ }{ }C\dfrac{ }{ }\dfrac{ }{ }\dfrac{ }{ }\dfrac{ }{ }}{ }\%\)