Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

1. \(n_{O_2}=\frac{V}{22,4}=\frac{6,72}{22,4}=0,3\left(mol\right)\)
2.
\(n_{CO_2}=\frac{m}{M}=\frac{4,4}{44}=0,1\left(mol\right)\)
\(n_{O_2}=\frac{m}{M}=\frac{3,2}{32}=0,1\left(mol\right)\)
\(V_{HC}=n.22,4=\left(0,1+0,1\right).22,4=4,48\left(l\right)\)

\(a.n_{CO_2}=\dfrac{m_{CO_2}}{M_{CO_2}}=\dfrac{4,4}{44}=0,1\left(mol\right)\\ \Rightarrow V_{CO_2}=n_{CO_2}.22,4=0,1.22,4=2,24\left(l\right)\\ n_{O_2}=\dfrac{m_{O_2}}{M_{O_2}}=\dfrac{3,2}{32}=0,1\left(mol\right)\)
\(\Rightarrow V_{O_2}=n_{O_2}.22,4=0,1.22,4=2,24\left(l\right)\)
\(n_{H_2}\) = 2mol
=>\(V_{H_2}\) = 2. 22,4 = 44,8l
\(n_{O_2}\) = 0,0875 mol
=>\(V_{O_2}\) = 0,0875 . 22,4 = 1,96l
\(n_{CO_2}\) = 0,5 mol
=>\(V_{CO_2}\) = 0,5 .22,4 = \(11,2\left(l\right)\)
\(n_{O_2}\) = \(0,2\left(mol\right)\)
=>\(V_{O_2}\) = 0,2 . 22,4 = \(4,48\left(l\right)\)
b) Ta có:
\(n_{NH_3}=\dfrac{6.10^{23}}{6.10^{23}}=1\left(mol\right)\\ n_{O_2}=\dfrac{3.10^{23}}{6.10^{23}}=0,5\left(mol\right)\\ \Sigma n_{hh}=1+0,5=1,5\left(mol\right)\\ V_{hh}=1,5.22,4=33,6\left(l\right)\)

\(n_{O_2}=\dfrac{3,2}{32}=0,1\left(mol\right)\)
=> Vhh = (0,1+0,15).22,4 = 5,6 (l)

\(n_{O_2}=\dfrac{12,8}{32}=0,4\left(mol\right)\\ n_{CO_2}=\dfrac{13,2}{44}=0,3\left(mol\right)\\ \Rightarrow V_{hh}=\left(0,4+0,3\right).22,4=15,68\left(l\right)\)

\(1,\\ a,m_{hh}=3.44+2.28=188(g)\\ b,m_{hh}=\dfrac{2,24}{22,4}.64+\dfrac{1,12}{22,4}.32=8(g)\\ 2,\\ a,V_{hh}=(\dfrac{4,4}{44}+\dfrac{0,4}{2}).22,4=6,72(l)\\ b,V_{hh}=(\dfrac{6.10^{23}}{6.10^{23}}+\dfrac{3.10^{23}}{6.10^{23}}).22,4=33,6(l)\)

nX = 0,672/22,4 = 0,03 (mol)
Gọi nN2 = a (mol); nO2 = b (mol)
a + b = 0,03
28a + 32b = 0,88
=> a = 0,02 (mol); b = 0,01 (mol)
%VN2 = 0,02/0,03 = 66,66%
%VO2 = 100% - 66,66% = 33,34%
M(X) = 0,88/0,03 = 88/3 (g/mol)
nX = 2,2 : 88/3 = 0,075 (mol)
VH2 = VX = 0,075 . 22,4 = 1,68 (l)

\(n_{NH_3}=\dfrac{5,1}{17}=0,3\left(mol\right);n_{O_2}=\dfrac{3,2}{32}=0,1\left(mol\right)\)
\(\Rightarrow n_{hh}=0,3+0,1=0,4\left(mol\right)\)
\(\Rightarrow V_{hh}=0,4.22,4=8,96\left(l\right)\)
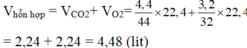

nO2 -m/M-4,4/32-0.1mol
VO2-n.22,4-0,1.22,4-2,24l
nO2=4,4/16=0,275(mol)
VO2=0,275.22,4=6,16 l