Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(\%Fe=\dfrac{56}{56.2+16.3}.100\%=35\%\\ \%O=\dfrac{16}{56.2+16.3}.100\%=10\%\)

Khối lượng mol của \(Fe2O3\) là :
\(M_{Fe_2}_{O_3}=56,2+16,3=160\left(g.mol\right)\)
\(\%Fe=\dfrac{56.2}{100}.100\%=70\%\)
\(\%O=100\%-70\%=30\%\)

\(M_{KMnO_4}=158(g/mol)\\ \%_{K}=\dfrac{39}{158}.100\%=24,68\%\\ \%_{Mn}=\dfrac{55}{158}.100\%=34,81\%\\ \%_O=100\%-24,68\%-34,81\%=40,51\%\)

\(\%m_{Ca}=\dfrac{40}{164}.100=24,39\left(\%\right)\\ \%m_N=\dfrac{28}{164}.100=17,07\left(\%\right)\\ \%m_O=\dfrac{48.2}{164}.100=58,54\left(\%\right)\)

Câu 2:
Trong 1 mol X: \(\left\{{}\begin{matrix}n_{Ag}=\dfrac{170.63,53\%}{108}=1\left(mol\right)\\n_N=\dfrac{170.8,23\%}{14}=1\left(mol\right)\\n_O=\dfrac{170\left(100\%-63,53\%-8,23\%\right)}{16}=3\left(mol\right)\end{matrix}\right.\)
Vậy CTHH của X là \(AgNO_3\)
Câu 1:
\(a,\%_{Fe}=\dfrac{56}{180}\cdot100\%=31,11\%\\ \%_N=\dfrac{14\cdot2}{180}\cdot10\%=15,56\%\\ \%_O=100\%-31,11\%-15,56\%=53,33\%\\ b,\%_{N\left(N_2O\right)}=\dfrac{14\cdot2}{44}\cdot100\%=63,63\%\\ \%_{O\left(N_2O\right)}=100\%-63,63\%=36,37\%\\ \%_{N\left(NO\right)}=\dfrac{14}{30}\cdot100\%=46,67\%\\ \%_{O\left(NO\right)}=100\%-46,67\%=53,33\%\\ \%_{O\left(NO_2\right)}=\dfrac{16\cdot2}{46}\cdot100\%=69,57\%\\ \%_{N\left(NO_2\right)}=100\%-69,57\%=30,43\%\)

\(\%m_S=\dfrac{32}{80}.100\%=40\%\)
\(\%m_O=\dfrac{3.16}{80}.100\%=60\%\)
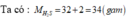
\(M_{H_2SO_4}=2+32+64=98\left(g\text{/}mol\right)\)
\(\%H=\dfrac{2}{98}\cdot100\%=2.04\%\)
\(\%S=\dfrac{32}{98}\cdot100\%=32.65\%\)
\(\%O=100-2.04-32.65=65.31\%\)
\(M_{H_2SO_4}=1.2+32+16.4=98\left(DvC\right)\\ \%H=\dfrac{1.2}{98}.100\%=2,04\%\\ \%S=\dfrac{32}{98}.100\%=32,6\%\\ \%O=100\%-2,04\%-32,6\%=65,36\%\)