Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) \(n_{CuO}=\dfrac{4}{80}=0,05\left(mol\right)\)
\(n_{HCl}=\dfrac{146.5\%}{36,5}=0,2\left(mol\right)\)
PTHH: CuO + 2HCl --> CuCl2 + H2O
Xét tỉ lệ: \(\dfrac{0,05}{1}< \dfrac{0,2}{2}\) => CuO hết, HCl dư
=> dd sau phản ứng chứa CuCl2, HCl dư
b)
PTHH: CuO + 2HCl --> CuCl2 + H2O
0,05-->0,1------>0,05
mdd sau pư = 4 + 146 = 150 (g)
\(\left\{{}\begin{matrix}C\%_{CuCl_2}=\dfrac{0,05.135}{150}.100\%=4,5\%\\C\%_{HCldư}=\dfrac{\left(0,2-0,1\right).36,5}{150}.100\%=2,433\%\end{matrix}\right.\)
b)
PTHH: NaOH + HCl --> NaCl + H2O
CuCl2 + 2NaOH --> 2NaCl + Cu(OH)2
0,05--------------------------->0,05
Cu(OH)2 --to--> CuO + H2O
0,05----------->0,05
=> \(a=m_{Cu\left(OH\right)_2}=0,05.98=4,9\left(g\right)\)
=> \(b=m_{CuO}=0,05.80=4\left(g\right)\)

Gọi kim loại cần tìm là A
a) PTHH: \(A+H_2O\rightarrow AOH+\dfrac{1}{2}H_2\uparrow\)
\(AOH+HCl\rightarrow ACl+H_2O\)
b) Ta có: \(n_{H_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\) \(\Rightarrow n_A=0,2mol\)
\(\Rightarrow M_A=\dfrac{7,8}{0,2}=39\) \(\Rightarrow\) Kim loại cần tìm là Kali
b) Ta có: \(\left\{{}\begin{matrix}n_{KCl}=0,2mol\\n_{HCl\left(pư\right)}=0,2mol\Rightarrow n_{HCl\left(dư\right)}=0,2\cdot20\%=0,04\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}m_{KCl}=0,2\cdot74,5=14,9\left(g\right)\\m_{HCl\left(dư\right)}=0,04\cdot36,5=1,46\left(g\right)\end{matrix}\right.\)
Mặt khác: \(m_{H_2}=2\cdot0,1=0,2\left(g\right)\)
\(\Rightarrow m_{dd}=m_K+m_{ddHCl}-m_{H_2}=7,8+\dfrac{0,24\cdot36,5}{10\%}-0,2=95,2\left(g\right)\)
\(\Rightarrow\left\{{}\begin{matrix}C\%_{KCl}=\dfrac{14,9}{95,2}\cdot100\%\approx15,65\%\\C\%_{HCl\left(dư\right)}=\dfrac{1,46}{95,2}\cdot100\%\approx1,53\%\end{matrix}\right.\)

a) \(n_{Al}=\dfrac{8,64}{27}=0,32\left(mol\right)\)
\(n_{HCl}=\dfrac{365.10\%}{36,5}=1\left(mol\right)\)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
Xét tỉ lệ \(\dfrac{0,32}{2}< \dfrac{1}{6}\) => Al hết, HCl dư
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
0,32-->0,96---->0,32--->0,48
=> \(V_{H_2}=0,48.22,4=10,752\left(l\right)\)
b) Trong Y chứa AlCl3 và HCl dư
\(m_{AlCl_3}=0,32.133,5=42,72\left(g\right)\)
c) mdd sau pư = 8,64 + 365 - 0,48.2 = 372,68 (g)
\(\left\{{}\begin{matrix}C\%\left(AlCl_3\right)=\dfrac{42,72}{372,68}.100\%=11,463\%\\C\%\left(HCldư\right)=\dfrac{\left(1-0,96\right).36,5}{372,68}.100\%=0,392\%\end{matrix}\right.\)

Đáp án B.
nFe = 0,2 mol; nZn = 0,1 mol
NaOH + H2S→NaHS + H2O
![]()

MnO 2 + HCl → MnCl 2 + Cl 2 + 2 H 2 O
Cl 2 + 2NaOH → NaCl + NaClO + H 2 O
n MnO 2 = 0,2 mol; n NaOH = 0,729 mol
Theo phương trình (1) ta có: n Cl 2 = n MnO 2 = 0,2 mol
Theo phương trình (2) ta có: 2 n Cl 2 < n NaOH ⇒ NaOH dư
Dung dịch A gồm: n NaCl = n NaClO = n Cl 2 = 0,2 mol
n NaOH dư = 0,729 – 2.0,2 = 0,329 mol
m dd A = m Cl 2 + m dd NaOH = 0,2.71 + 145,8 = 160g
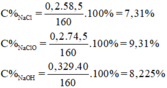

Ta có: \(n_{SO_2}=\dfrac{12,395}{24,79}=0,5\left(mol\right)\)
BTNT S, có: nH2SO4 = nSO3 = nSO2 = 0,5 (mol)
Mà: mH2SO4 (ban đầu) = 210.10% = 21 (g)
⇒ mH2SO4 (trong X) = 21 + 0,5.98 = 70 (g)
Có: m dd X = 210 + mSO3 = 210 + 0,5.80 = 250 (g)
\(\Rightarrow C\%_{H_2SO_4}=\dfrac{70}{250}.100\%=28\%\)




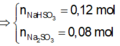
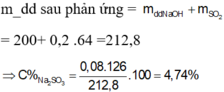
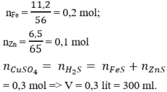
\(MnO_2+4HCl\rightarrow MnCl_2+Cl_2+2H_2O\)
Ta có :
\(n_{MnO2}=\frac{10,44}{87}=0,12\left(mol\right)\)
\(n_{Cl2}=n_{MnO2}=0,12\left(mol\right)\)
\(n_{NaOH}=\frac{60.20\%}{40}=0,3\left(mol\right)\)
\(Cl_2+2NaOH\rightarrow NaCl+NaClO+H_2O\)
\(n_{NaOH.trong.Y}=0,3-0,12.2=0,06\left(mol\right)\)
\(n_{NaCl}=n_{NaClO}=0,12\left(mol\right)\)
\(m_{dd\left(Spu\right)}=0,12.71+60=68,52\left(g\right)\)
\(\Rightarrow\left\{{}\begin{matrix}C\%_{NaOH}=3,5\%\\C\%_{NaCl}=10,25\%\\C\%_{NaClO}=13,05\%\end{matrix}\right.\)