Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{NaOH}=0.4\cdot0.1=0.04\left(mol\right)\)
TH1 : NaOH dư
\(n_{Na_2S}=\dfrac{1.9}{78}=\dfrac{19}{780}\left(mol\right)\)
\(2NaOH+H_2S\rightarrow Na_2S+2H_2O\)
\(n_{NaOH}=\dfrac{19}{780}\cdot2=0.048>0.04\left(L\right)\)
TH2 : Tạo cả 2 muối , NaOH phản ứng đủ
\(n_{Na_2S}=a\left(mol\right),n_{NaHS}=b\left(mol\right)\)
\(m=78a+56b=1.9\left(g\right)\left(1\right)\)
\(n_{NaOH}=2a+b=0.04\left(mol\right)\left(2\right)\)
\(\left(1\right),\left(2\right):a=0.01,b=0.02\)
\(V_{H_2S}=\left(0.01+0.02\right)\cdot22.4=0.672\left(l\right)=672\left(ml\right)\)

\(n_{Na_2S}=n_{NaHS}=a\left(mol\right)\)
\(n_{NaOH}=2a+a=3a=0.03\left(mol\right)\)
\(\Rightarrow a=0.01\)
\(V=\left(0.01+0.01\right)\cdot22.4=0.448\left(l\right)\)

\(n_{H_2S}=\dfrac{0.672}{22.4}=0.03\left(mol\right)\)
\(a.\)
\(n_{NaOH}=0.75\cdot0.1=0.075\left(mol\right)\)
\(T=\dfrac{0.075}{0.03}=2.5>2\)
=> Tạo muối trung hòa
\(2NaOH+H_2S\rightarrow Na_2S+H_2O\)
\(0.06............0.03.........0.03\)
\(m_{Na_2S}=0.03\cdot78=2.34\left(g\right)\)
\(b.\)
\(n_{NaOH}=0.42\cdot0.1=0.042\left(mol\right)\)
\(T=\dfrac{0.042}{0.03}=1.4\)
=> Tạo 2 muối
\(n_{Na_2S}=a\left(mol\right),n_{NaHS}=b\left(mol\right)\)
\(\left\{{}\begin{matrix}2a+b=0.042\\a+b=0.03\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}a=0.012\\b=0.018\end{matrix}\right.\)
\(m_{Muối}=0.012\cdot78+0.018\cdot56=1.944\left(g\right)\)

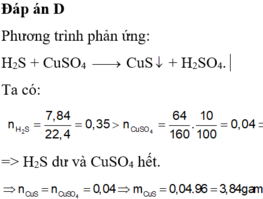
Đáp án D.
![]()
H2S + CuSO4 → CuS + H2SO4
0,35 0,04 0,04 (H2S dư)
m = 0,04.(64+32) = 3,84 (g)

\(n_{H_2S}=0,08\left(mol\right),n_{OH^-}=0,1.0,05+0,1.0,08=0,013\left(mol\right)\)
T=\(\dfrac{0,013}{0,08}=0,1625\)=> Tạo 1 muối HS- , H2S dư
Muối gồm KHS và NaHS
=> \(m_{muối}=0,1.0,05.56+0,1.0,08.72=0,856\left(g\right)\)

\(n_{H_2S}=\dfrac{0.448}{22.4}=0.02\left(mol\right)\)
\(n_{NaOH}=0.1\cdot0.5=0.05\left(mol\right)\)
\(T=\dfrac{0.05}{0.02}=2.5>2\)
\(2NaOH+H_2S\rightarrow Na_2S+H_2O\)
\(0.04........0.02..............0.02\)
\(n_{Na_2S}=0.02\left(mol\right)\)
\(n_{NaOH\left(dư\right)}=0.05-0.04=0.01\left(mol\right)\)
\(n_{NaOH}=0.24\cdot0.1=0.024\left(mol\right)\)
\(T=\dfrac{0.024}{0.02}=1.2\)
=> Tạo 2 muối
\(n_{Na_2S}=a\left(mol\right),n_{NaHS}=b\left(mol\right)\)
\(\left\{{}\begin{matrix}2a+b=0.024\\a+b=0.02\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}a=0.004\\b=0.016\end{matrix}\right.\)

Đáp án A
nX = 0,4 (mol)
n↓ = nPbS = 0,1 (mol)
Pb(NO3)2 + H2S →PbS + 2HNO3
![]()
nPb(NO3)2 = 0,04(mol)
nH2S=0,03(mol)
PTHH: Pb(NO3)2 + H2S -> PbS (kt) + 2 HNO3
nPbS=nH2S=0,03 (mol) (H2S hết, Pb(NO3)2 dư)
=> mPbS= 239. 0,03=7,17(g)
KL kết tủa 7,17 gam á em nha!