Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

Bài 1 :
\(n_{Mg}=\frac{2,4}{24}=0,1\left(mol\right)\) ; \(n_{HCl}=\frac{3,65}{36,5}=0,1\left(mol\right)\)
PTHH : \(Mg+2HCl-->MgCl_2+H_2\)
Ta thấy : \(\frac{n_{HCl}}{2}< n_{Mg}\left(0,05< 0,1\right)\)=> Spu Mg còn dư
Theo pthh : \(n_{H_2}=n_{MgCl_2}=n_{Mg\left(pứ\right)}=\frac{1}{2}n_{HCl}=0,05\left(mol\right)\)
=> \(\hept{\begin{cases}m_{Mg\left(dư\right)}=\left(0,1-0,05\right)\cdot24=1,2\left(g\right)\\m_{MgCl_2}=95\cdot0,05=4,75\left(g\right)\\V_{H_2}=0,05\cdot22,4=1,12\left(l\right)\end{cases}}\)
Bài 2 :
\(n_{Al}=\frac{5,4}{27}=0,2\left(mol\right)\) ; \(n_{H_2SO_4}=\frac{14,7}{98}=0,15\left(mol\right)\)
PTHH : \(2Al+3H_2SO_4-->Al_2\left(SO_4\right)_3+3H_2\)
Ta thấy : \(\frac{n_{Al}}{2}>\frac{n_{H_2SO_4}}{3}\left(0,1>0,05\right)\) => Spu Al còn dư
Theo pthh : \(n_{Al\left(pứ\right)}=\frac{2}{3}n_{H_2SO_4}=0,1\left(mol\right)\)
\(n_{Al_2\left(SO_4\right)_3}=\frac{1}{3}n_{H_2SO_4}=0,05\left(mol\right)\)
\(n_{H_2}=n_{H_2SO_4}=0,15\left(mol\right)\)
=> \(\hept{\begin{cases}m_{Al\left(dư\right)}=\left(0,2-0,1\right)\cdot27=2,7\left(g\right)\\m_{Al_2\left(SO_4\right)_3}=342\cdot0,05=17,1\left(g\right)\\V_{H_2}=0,15\cdot22,4=3,36\left(l\right)\end{cases}}\)
Bài 3 :
\(n_{H_2}=\frac{4,704}{22,4}=0,21\left(mol\right)\)
PTHH : \(2M+6HCl-->2MCl_3+3H_2\)
Theo pthh : \(n_M=\frac{2}{3}n_{H_2}=0,14\left(mol\right)\)
=> \(\frac{3,78}{M_M}=0,14\)
=> \(M_M=27\) (g/mol)
=> Kim loại M là Nhôm (Al)
Bài 4 :
\(n_P=\frac{6,2}{31}=0,2\left(mol\right)\)
PTHH : \(2KMnO_4-t^o->K_2MnO_4+MnO_2+O_2\) (1)
\(4P+5O_2-t^o->2P_2O_5\) (2)
Theo pthh (1); \(n_{O_2}=\frac{1}{2}n_{KMnO_4}=0,2\left(mol\right)\)
Xét pứ (2) , thấy : \(\frac{n_P}{4}>\frac{n_{O2}}{5}\left(0,05>0,04\right)\) => spu photpho còn dư
Theo pthh (2) : \(n_{P_2O_5}=\frac{2}{5}n_{O_2}=0,08\left(mol\right)\)
=> \(m_{P_2O_5}=0,08\cdot142=11,36\left(g\right)\)

PTHH: \(CH_4+2O_2\rightarrow^{t^o}CO_2+2H_2O\)
\(2C_4H_{10}+13O_2\rightarrow^{t^o}8CO_2+10H_2O\)
Đặt \(\hept{\begin{cases}x\left(mol\right)=CH_4\\y\left(mol\right)=C_4H_{10}\end{cases}}\), có hệ phương trình sau
\(\hept{\begin{cases}16x+58y=7,4\\44x+176y=22\end{cases}}\)
\(\Leftrightarrow x=y=0,1\)
Dưa vào PTHH 1: \(n_{O_2}=2x=0,1.2=0,2mol\)
Dựa vào PTHH 2: \(n_{O_2}=\frac{13.0,1}{2}=0,65mol\)
Có tổng số mol của \(O_2\) là: \(0,2+0,65=0,85mol\)
\(\rightarrow m_{O_2}=32.0,85=27,2g\)

a) \(n_{O_2}=\dfrac{11,2.20\%}{22,4}=0,1\left(mol\right)\)
PTHH: 2Mg + O2 --to--> 2MgO
0,2<--0,1--------->0,2
=> mMg = 0,2.24 = 4,8 (g)
b) nMgO = 0,2.40 = 8 (g)

\(n_{Mg}=\dfrac{2,4}{24}=0,1\left(mol\right)\\ a,2Mg+O_2\rightarrow\left(t^o\right)2MgO\\ n_{O_2}=\dfrac{0,1}{2}=0,05\left(mol\right)\\ V_{O_2\left(đktc\right)}=0,05.22,4=1,12\left(l\right)\\ b,2KClO_3\rightarrow\left(t^o\right)2KCl+3O_2\\ n_{KClO_3}=\dfrac{0,05.2}{3}=\dfrac{1}{30}\left(mol\right)\\ \Rightarrow m_{KClO_3}=\dfrac{122,5}{30}=\dfrac{49}{12}\left(g\right)\)

\(n_{O_2}=\dfrac{V}{22,4}=\dfrac{11,2}{22,4}=0,5mol\)
PTHH:
\(2Mg+O_2->2MgO\)
2 : 1 : 2 mol
1 : 0,5 : 1 mol
\(m_{Mg}=n.M=1.24=24g\)
\(m_{MgO}=n.M=1.\left(24+16\right)=40g\)
nO2 = 11,2/22,4 = 0,5 (mol)
PTHH: 2Mg + O2 -> (t°) 2MgO
Mol: 1 <--- 0,5 ---> 1
mMg = 1 . 24 = 24 (g)
mMgO = 1 . 40 = 40 (g)

a, \(2Zn+O_2\underrightarrow{t^o}2ZnO\)
b, \(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\)
Theo PT: \(n_{O_2}=\dfrac{1}{2}n_{Zn}=0,1\left(mol\right)\Rightarrow V_{O_2}=0,1.22,4=2,24\left(l\right)\)
\(\Rightarrow V_{kk}=5V_{O_2}=11,2\left(l\right)\)
c, \(2KMnO_4\underrightarrow{t^o}K_2MnO_4+MnO_2+O_2\)
Theo PT: \(n_{KMnO_4}=2n_{O_2}=0,2\left(mol\right)\Rightarrow m_{KMnO_4}=0,2.158=31,6\left(g\right)\)

a)
\(n_{Mg}=\dfrac{12}{24}=0,5\left(mol\right)\)
PTHH: 2Mg + O2 --to--> 2MgO
______0,5-->0,25---->0,5
=> VO2 = 0,25.22,4 = 5,6 (l)
=> mMgO = 0,5.40 = 20 (g)
b)
\(n_{O_2}=0,25=>n_{CO_2}=0,25\)
=> mCO2 = 0,25.44 = 11 (g)

a) $2Mg + O_2 \xrightarrow{t^o} 2MgO$
Theo PTHH : n MgO = n Mg = 2,4/24 = 0,1(mol)
=> m MgO = 0,1.40 = 4(gam)
b) n O2 = 1/2 n Mg = 0,05(mol)
=> V O2 = 0,05.22,4 = 1,12(lít)
=> V không khí = 5V O2 = 1,12.5 = 5,6(lít)
c) $Mg + 2HCl \to MgCl_2 + H_2$
n HCl = 200.15%/36,5 = 60/73(mol)
Ta thấy :
n Mg / 1 = 0,1 < n HCl / 2 = 30/73 suy ra HCl dư
n H2 = n Mg = 0,1(mol)
=> m dd sau pư = 2,4 + 200 - 0,1.2 = 202,2(gam)
Vậy :
C% MgCl2 = 0,1.95/202,2 .100% = 4,7%
C% HCl = (60/73 - 0,1.2).36,5/202,2 .100% = 11,23%

Theo gt ta có: $n_{H_2}=0,75(mol)$
a, $2H_2+O_2\rightarrow 2H_2O$
Ta có: $n_{O_2}=0,5.n_{H_2}=0,375(mol)\Rightarrow V_{O_2}=8,4(l)\Rightarrow V_{kk}=42(l)$
b, $2KMnO_4\rightarrow K_2MnO_4+MnO_2+O_2$
Ta có: $n_{KMnO_4}=2.n_{O_2}=0,75(mol)\Rightarrow m_{KMnO_4}=118,5(g)$
a)
\(2H_2 + O_2 \xrightarrow{t^o} 2H_2O\\ V_{O_2} = \dfrac{V_{H_2}}{2} = \dfrac{16,8}{2} = 8,4(lít)\\ V_{không\ khí} = \dfrac{8,4}{20\%} = 42(lít)\)
b)
\(n_{O_2} = \dfrac{8,4}{22,4} = 0,375(mol)\\ 2KMnO_4 \xrightarrow{t^o} K_2MnO_4 + MnO_2 + O_2\\ n_{KMnO_4} = 2n_{O_2} = 0,75(mol)\\ \Rightarrow m_{KMnO_4} = 0,75.158 = 118,5(gam)\\ 2KClO_3 \xrightarrow{t^o} 2KCl + 3O_2\\ n_{KClO_3} = \dfrac{2}{3}n_{O_2} = 0,25(mol)\\ \Rightarrow m_{KClO_3} = 0,25.122,5 = 30,625(gam)\)

a)
\(4P + 5O_2 \xrightarrow{t^o} 2P_2O_5\)
Sản phẩm : Điphotpho pentaoxit.
b)
\(n_P = \dfrac{6,2}{31} = 0,2(mol)\\ \Rightarrow n_{P_2O_5} = \dfrac{1}{2}n_P = 0,1(mol)\\ \Rightarrow m_{P_2O_5} = 0,1.142 = 14,2(gam)\)
c)
\(n_{O_2} = \dfrac{5}{4}n_P = 0,125(mol)\\ \Rightarrow V_{O_2} = 0,125.22,4 = 2,8(lít)\)
d)
\(V_{không\ khí} = \dfrac{2,8}{20\%} = 14(lít)\)
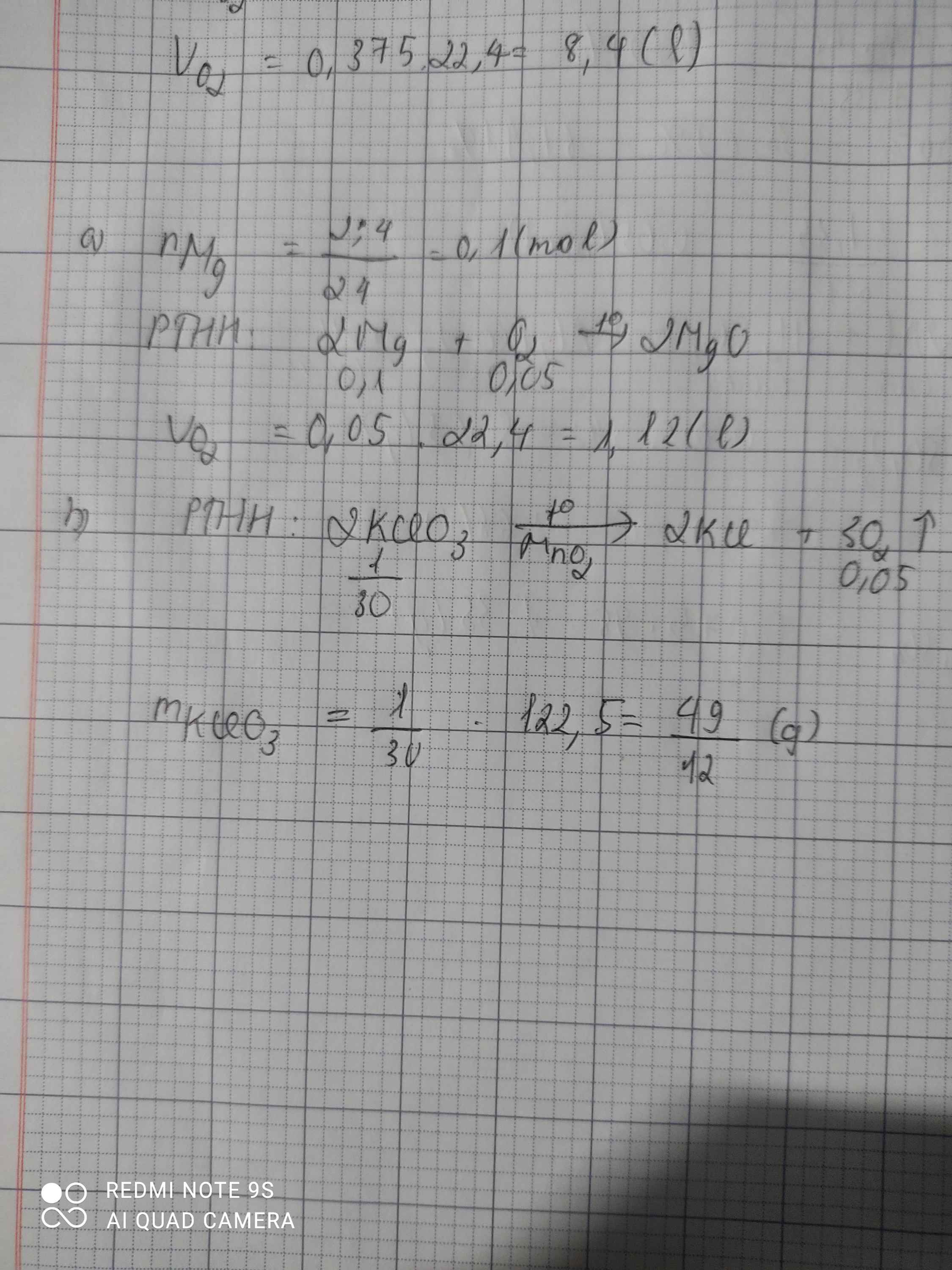
\(a,n_{MgO}=\dfrac{8}{40}=0,2\left(mol\right)\)
PTHH: 2Mg + O2 --to--> 2MgO
0,2---<0,1<--------0,2
Mg + 2HCl ---> MgCl2 + H2
0,2--->0,4
=> \(\left\{{}\begin{matrix}b,m_{Mg}=0,2.24=4,8\left(g\right)\\c,V_{kk}=0,1.5.22,4=11,2\left(l\right)\\d,m_{ddHCl}=\dfrac{0,4.36,5}{10\%}=146\left(g\right)\end{matrix}\right.\)