
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


\(n_{H_2}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\)
PTHH: Mg + 2CH3COOH ---> (CH3COO)2Mg + H2
0,15<-------------------------------------------0,15
\(\Rightarrow\left\{{}\begin{matrix}m_{Mg}=0,15.24=3,6\left(g\right)\\m_{MgO}=7,6-3,6=4\left(g\right)\end{matrix}\right.\\ \Rightarrow B\)

\(n_{NaOH}=0,1.0,15=0,03\left(mol\right)\)
PTHH: CH3COOH + NaOH ---> CH3COONa + H2O
0,03<----------0,03
\(\Rightarrow C_{M\left(CH_3COOH\right)}=\dfrac{0,03}{0,1}=0,3M\\ \Rightarrow B\)

C5:
nC2H5OH = 8,2 / 46 = 0,2 (mol)
C2H5OH + 3O2-- (t^o)-- > 2CO2 + 3H2O
VCO2 = 0,4 . 22,4 = 8,96 (l)
VO2 = 0,6.22,4=13,44 (l)

Sửa đề: 7,2% → 7,3%
a, \(Fe+2HCl\rightarrow FeCl_2+H_2\)
b, \(n_{Fe}=\dfrac{2,8}{56}=0,05\left(mol\right)\)
\(m_{HCl}=100.7,3\%=7,3\left(g\right)\Rightarrow n_{HCl}=\dfrac{7,3}{36,5}=0,2\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{0,05}{1}< \dfrac{0,2}{2}\), ta được HCl dư.
Theo PT: \(\left\{{}\begin{matrix}n_{FeCl_2}=n_{H_2}=n_{Fe}=0,05\left(mol\right)\\n_{HCl\left(pư\right)}=2n_{Fe}=0,1\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow m_{FeCl_2}=0,05.127=6,35\left(g\right)\)
\(m_{H_2}=0,05.2=0,1\left(g\right)\)
\(n_{HCl\left(dư\right)}=0,2-0,1=0,1\left(mol\right)\Rightarrow m_{HCl\left(dư\right)}=0,1.36,5=3,65\left(g\right)\)



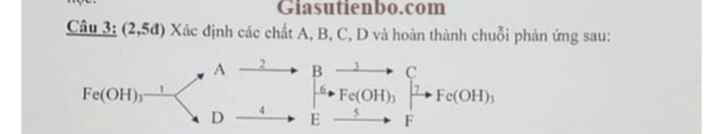
\(A:Fe_2O_3\\ B:FeCl_3\\ C:Fe(NO_3)_3\\ D:Fe_2(SO_4)_3\\ E:Na_2SO_4\\ F:NaOH\)
Từ đó ta có các PTHH tương ứng là:
\((1)Fe(OH)_3\xrightarrow{t^o}Fe_2O_3+3H_2O\\ (2)Fe_2O_3+6HCl\to 2FeCl_3+3H_2O\\ (3)FeCl_3+3AgNO_3\to Fe(NO_3)_3+3AgCl\downarrow\\ (4)Fe_2(SO_4)_3+6NaOH\to 2Fe(OH)_3\downarrow+3Na_2SO_4\\ (5)Na_2SO_4+Ba(OH)_2\to BaSO_4\downarrow+2NaOH\\ (6)FeCl_3+3NaOH\to Fe(OH)_3+3NaCl\\ (7)\begin{cases} Fe(NO_3)_3+3NaOH\to Fe(OH)_3\downarrow+3NaNO_3\\ NaOH+FeCl_3\to Fe(OH)_3+3NaCl \end{cases}\)

\(a,n_{CO_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\\ Ba\left(OH\right)_2+CO_2\rightarrow BaCO_3+H_2O\\ 0,1.......0,1.......0,1\left(mol\right)\\ C_{MddBa\left(OH\right)_2}=\dfrac{0,1}{0,2}=0,5\left(M\right)\\ b,m_{\downarrow}=m_{BaCO_3}=197.0,1=19,7\left(g\right)\)
\(a,PTHH:CO_2+Ba\left(OH\right)_2\rightarrow BaCO_3\downarrow+H_2O\\ \Rightarrow n_{Ba\left(OH\right)_2}=n_{CO_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\\ \Rightarrow C_{M_{Ba\left(OH\right)_2}}=\dfrac{0,1}{0,2}=0,5M\\ b,n_{BaCO_3}=n_{CO_2}=0,1\left(mol\right)\\ \Rightarrow m_{BaCO_3}=0,1\cdot197=19,7\left(g\right)\)








Độ rượu (sau khi pha) là: \(\dfrac{\dfrac{20.90}{100}}{20+25}.100=40^o\)
=> A