Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_P=\dfrac{3,1}{31}=0,1\left(mol\right)\\ n_{O_2}=\dfrac{3,2}{32}=0,1\left(mol\right)\\ pthh:4P+5O_2\underrightarrow{t^o}2P_2O_5\\ LTL:\dfrac{0,1}{4}>\dfrac{0,1}{5}\)
=> P dư
\(n_{P\left(p\text{ư}\right)}=\dfrac{4}{5}n_{O_2}=0,08\left(mol\right)\\
m_{P\left(d\right)}=\left(0,1-0,08\right).31=0,62\left(g\right)\)
\(n_P=\dfrac{3,1}{31}=0,1\left(mol\right)\\ n_{O_2}=\dfrac{3,2}{32}=0,1\left(mol\right)\)
PTHH: \(4P+5O_2\xrightarrow[]{t^o}2P_2O_5\)
bđ 0,1 0,1
pư 0,08 0,1
spư 0,02 0
=> P dư
\(m_{P\left(dư\right)}=0,02.31=0,62\left(g\right)\)

nP = 2,48/31 = 0,08 (mol)
PTHH: 4P + 5O2 -> (t°) 2P2O5
Mol: 0,08 ---> 0,1 ---> 0,04
mP2O5 = 0,04 . 142 = 5,68 (g)
b) nO2 = 4/32 = 0,125 (mol)
So sánh: 0,125 > 0,1 => O2 dư
nO2 (dư) = 0,125 - 0,1 = 0,025 (mol)
mO2 (dư) = 0,025 . 32 = 0,8 (g)

\(n_P=\dfrac{12,4}{31}=0,4\left(mol\right)\\
pthh:4P+5O_2\underrightarrow{t^o}2P_2O_5\)
0,4 0,2
\(m_{P_2O_5}=142.0,2=28,4g\)
\(n_{O_2}=\dfrac{17}{32}=0,53\left(mol\right)\)
\(pthh:4P+5O_2\underrightarrow{t^o}2P_2O_5\\
LTL:\dfrac{0,4}{4}< \dfrac{0,53}{5}\)
=> O2 dư
\(n_{O_2\left(p\text{ư}\right)}=\dfrac{5}{4}n_P=0,5\left(mol\right)\\
m_{O_2\left(d\right)}=\left(0,53-0,5\right).32=0,96g\)
`4P + 5O_2` $\xrightarrow[]{t^o}$ `2P_2 O_5`
`0,4` `0,5` `0,2` `(mol)`
`n_P = [ 12,4 ] / 31 = 0,4 (mol)`
`a) m_[P_2 O_5] = 0,2 . 142 = 28,4 (g)`
`b) n_[O_2] = 17 / 32 = 0,53125 (mol)`
Ta có: `[ 0,4 ] / 4 < [ 0,53125 ] / 5`
`->O_2` dư
`=> m_[O_2 (dư)] = ( 0,53125 - 0,5 ) . 32 = 1(g)`

nP = 6.2/31 = 0.2 (mol)
nO2 = 6.72/22.4 = 0.3 (mol)
4P + 5O2 -to-> 2P2O5
0.2___0.25_____0.1
mO2 dư = ( 0.3 - 0.25) * 32 = 1.6(g)
mP2O5 = 0.1*142 = 14.2 (g)
Ta có: \(n_P=\dfrac{6.2}{31}=0.29mol\)
\(n_{O_2}=\dfrac{6.72}{22.4}=0.3mol\)
PTHH:
\(4P+5O_2\underrightarrow{t^o}2P_2O_5\)
ta có:
\(\left\{{}\begin{matrix}\dfrac{n_{P\left(bra\right)}}{nP_{\left(pthh\right)}}=\dfrac{0.2}{4}=0.05\\\dfrac{n_{O_2\left(bra\right)}}{n_{O_2}\left(pthh\right)}=\dfrac{0.3}{5}=0.06\end{matrix}\right.\)
=> \(O_2\) dư
\(4P+5O_2\underrightarrow{t^o}2P_2O_5\)
4 ----------->2
0.2---------->0.1=nP2O5
=>\(m_{P_2O_5}=142.0.1=14.2\left(g\right)\)

nP=\(\dfrac{62}{31}\)=0,2(mol)
nO2=\(\dfrac{7,84}{22,4}\)=0,35(mol)
PTHH:4P+5O2to→2P2O5
tpứ: 0,2 0,35
pứ: 0,2 0,25 0,1
spứ: 0 0,1 0,1
a)chất còn dư là oxi
mO2dư=0,1.32=3,2(g)
b)mP2O5=n.M=0,1.142=14,2(g)
\(a.n_P=0,2\left(mol\right);n_{O_2}=0,35\left(mol\right)\\ 4P+5O_2-^{t^o}\rightarrow2P_2O_5\\ LTL:\dfrac{0,2}{4}< \dfrac{0,35}{5}\\ \Rightarrow SauphảnứngO_2dư\\ n_{O_2\left(pứ\right)}=\dfrac{5}{4}n_P=0,25\left(mol\right)\\ \Rightarrow m_{P\left(dư\right)}=\left(0,35-0,25\right).32=3,2\left(g\right)\\ b.n_{P_2O_5}=\dfrac{1}{2}n_P=0,1\left(mol\right)\\ \Rightarrow m_{P_2O_5}=0,1.142=14,2\left(g\right)\)

$a) 4P + 5O_2 \xrightarrow{t^o} 2P_2O_5$
$n_P = \dfrac{6,2}{31} = 0,2(mol) ; n_{O_2} = \dfrac{7,84}{22,4} = 0,35(mol)$
$n_P : 4 = 0,05 < n_{O_2} :5 = 0,07$ nên $O_2$ dư
$n_{O_2\ pư} = \dfrac{5}{4}n_P = 0,25(mol)$
$\Rightarrow m_{O_2\ dư} = (0,35 - 0,25).32 = 3,2(gam)$
c) $n_{P_2O_5} = \dfrac{1}{2}n_P = 0,1(mol)$
$m_{P_2O_5} = 0,1.142 = 14,2(gam)$
\(n_P=\dfrac{6,2}{31}=0,2\left(mol\right)\\ n_{O_2}=\dfrac{7,84}{22,4}=0,35\left(mol\right)\\a, 4P+5O_2\rightarrow\left(t^o\right)2P_2O_5\\ V\text{ì}:\dfrac{0,35}{5}>\dfrac{0,2}{4}\Rightarrow O_2d\text{ư}\\ n_{O_2\left(d\text{ư}\right)}=0,35-\dfrac{5}{4}.0,2=0,1\left(mol\right)\\b, m_{O_2\left(d\text{ư}\right)}=0,1.32=3,2\left(g\right)\\ c,n_{P_2O_5}=\dfrac{n_P}{2}=\dfrac{0,2}{2}=0,1\left(mol\right)\\ m_{r\text{ắn}}=m_{P_2O_5}=142.0,1=14,2\left(g\right)\)

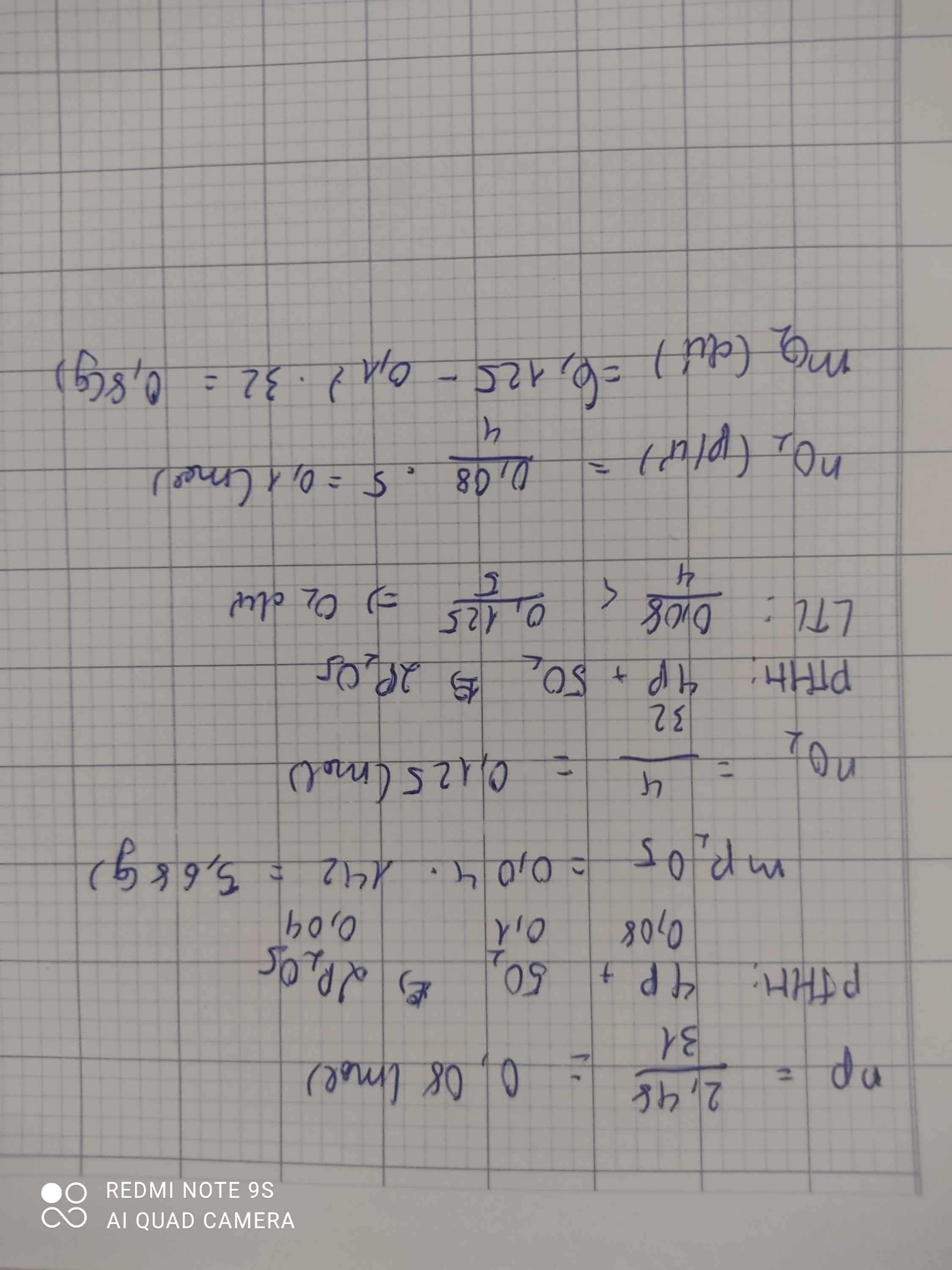
\(n_P=\dfrac{3,1}{31}=0,1\left(mol\right)\\ n_{O_2}=\dfrac{3,2}{32}=0,1\left(mol\right)\\ pthh:4P+5O_2\underrightarrow{t^o}2P_2O_5\\ LTL:\dfrac{0,1}{4}>\dfrac{0,1}{5}\)
P dư
\(n_{P\left(p\text{ư}\right)}=\dfrac{4}{5}n_{O_2}=0,08\left(mol\right)\\ m_{P\left(d\right)}=0,08.31=0,62\left(g\right)\\ n_{P_2O_5}=\dfrac{2}{5}n_{O_2}=0,04\left(mol\right)\\ m_{P_2O_5}=0,04.142=5,68\left(g\right)\\ m_{sp}=0,62+5,68=6,3\left(g\right)\)