Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) \(n_{CH_3COOH}=\dfrac{120.20\%}{60}=0,4\left(mol\right)\)
\(n_{Na_2CO_3}=\dfrac{53.30\%}{106}=0,15\left(mol\right)\)
PTHH: Na2CO3 + 2CH3COOH --> 2CH3COONa + CO2 + H2O
Xét tỉ lệ: \(\dfrac{0,15}{1}< \dfrac{0,4}{2}\) => Na2CO3 hết, CH3COOH dư
PTHH: Na2CO3 + 2CH3COOH --> 2CH3COONa + CO2 + H2O
0,15-------->0,3-------------->0,3------->0,15
=> \(m_{CH_3COONa}=0,3.82=24,6\left(g\right)\)
b) mdd sau pư = 120 + 53 - 0,15.44 = 166,4 (g)
=> \(C\%=\dfrac{24,6}{166,4}.100\%=14,78\text{%}\)

PTHH: \(CH_3COOH+NaOH\rightarrow CH_3COONa+H_2O\)
Ta có: \(n_{NaOH}=\dfrac{30\cdot20\%}{40}=0,15\left(mol\right)=n_{CH_3COONa}=n_{CH_3COOH}\)
\(\Rightarrow\left\{{}\begin{matrix}m_{CH_3COONa}=0,15\cdot82=12,3\left(g\right)\\C_{M_{CH_3COOH}}=\dfrac{0,15}{0,5}=0,3\left(M\right)\end{matrix}\right.\)

\(n_{CH_3COOH}=\dfrac{50.12\%}{60}=0,1mol\)
\(2CH_3COOH+Na_2CO_3\rightarrow2CH_3COONa+CO_2+H_2O\)
0,1 0,05 ( mol )
\(m_{Na_2CO_3}=0,05.106=5,3g\)
\(m_{dd_{Na_2CO_3}}=\dfrac{5,3}{8,4\%}=63,09g\)
\(m_{CH_3COOH}=\dfrac{12.50}{100}=6\left(g\right)\\ \rightarrow n_{CH_3COOH}=\dfrac{6}{60}=0,1\left(mol\right)\)
PTHH: 2CH3COOH + Na2CO3 ---> 2CH3COONa + CO2 + H2O
0,1------------>0,05
=> \(m_{ddNa_2CO_3}=\dfrac{0,05.106}{8,4\%}=63,1\left(g\right)\)

\(n_{CuO}=\dfrac{8}{80}=0,1\left(mol\right)\)
a) Pt : \(CuO+2HCl\rightarrow CuCl_2+H_2O|\)
1 2 1 1
0,1 0,2 0,1
b) \(n_{HCl}=\dfrac{0,1.2}{1}=0,2\left(mol\right)\)
⇒ \(m_{HCl}=0,2.36,5=7,3\left(g\right)\)
\(C_{ddHCl}=\dfrac{7,3.100}{200}=3,65\)0/0
c) \(n_{CuCl2}=\dfrac{0,2.1}{2}=0,1\left(mol\right)\)
⇒ \(m_{CuCl2}=0,1.135=13,5\left(g\right)\)
Chúc bạn học tốt
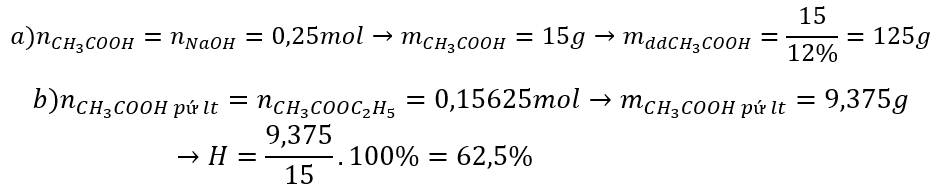
PTHH: CH3COOH + NaHCO3 ➞ CH3COONa + H2O + CO2
Ta có: mNAHCO3=(200.84)/100= 16.8 gam
nNAHCO3= 16.8/84= 0.2 mol
mCH3COOH= 0.2*60= 12 gam
Câu a) mddCH3COOH= (12*100)/6= 200 gam
Câu b) mddCH3COONA= mddCH3COOH + mdd NAHCO3= 200+200=400 gam
mCH3COONA= 0.2*82=16.4 gam
C%dd CH3COONA= (16.4*100)/400= 4.1%
Chúc bạn học tốt