Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) C2H4 + Br2 --> C2H4Br2
b) \(n_{Br_2}=\dfrac{24}{160}=0,15\left(mol\right)\)
PTHH: C2H4 + Br2 --> C2H4Br2
0,15<--0,15----->0,15
=> \(\%V_{C_2H_4}=\dfrac{0,15.22,4}{7,84}.100\%=42,857\%\)
=> \(\%V_{CH_4}=\dfrac{7,84-0,15.22,4}{7,84}.100\%=57,143\%\)
c) mC2H4Br2 = 0,15.188 = 28,2 (g)

a) C2H4 + Br2 --> C2H4Br2
b) nBr2 = 0,2.0,2 = 0,04 (mol)
PTHH: C2H4 + Br2 --> C2H4Br2
0,04<--0,04
=> \(m_{C_2H_4}=0,04.28=1,12\left(g\right)\)
\(m_{CH_4}=n_{CH_4}.M_{CH_4}=\left(\dfrac{1,12}{22,4}-0,04\right).16=0,16\left(g\right)\)
c) \(\left\{{}\begin{matrix}\%V_{C_2H_4}=\dfrac{0,04.22,4}{1,12}.100\%=80\%\\\%V_{CH_4}=100\%-80\%=20\%\end{matrix}\right.\)

a, \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
\(C_2H_2+2Br_2\rightarrow C_2H_2Br_4\)
Ta có: \(n_{C_2H_4}+n_{C_2H_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\left(1\right)\)
Theo PT: \(n_{Br_2}=n_{C_2H_4}+2n_{C_2H_2}=\dfrac{48}{160}=0,3\left(mol\right)\left(2\right)\)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}x=0,2\left(mol\right)\\y=0,05\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%V_{C_2H_4}=\dfrac{0,2.22,4}{5,6}.100\%=80\%\\\%V_{C_2H_2}=20\%\end{matrix}\right.\)
b, \(V_{ddBr_2}=\dfrac{0,3}{0,25}=1,2\left(M\right)\)

Ta có: \(n_{Br_2}=\dfrac{6}{160}=0,0375\left(mol\right)\)
PT: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
Theo PT: \(n_{C_2H_4}=n_{Br_2}=0,0375\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}\%V_{C_2H_4}=\dfrac{0,0375.22,4}{6,72}.100\%=12,5\%\\\%V_{CH_4}=87,5\%\end{matrix}\right.\)

a, Ta có: \(n_{CH_4}+n_{C_2H_4}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\left(1\right)\)
\(16n_{CH_4}+28n_{C_2H_4}=3\left(2\right)\)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}n_{CH_4}=0,1\left(mol\right)\\n_{C_2H_4}=0,05\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{CH_4}=\dfrac{0,1.16}{3}.100\%\approx53,33\%\\\%m_{C_2H_4}\approx46,67\%\end{matrix}\right.\)
- Ở cùng điều kiện nhiệt độ và áp suất, % số mol cũng là %V.
\(\Rightarrow\left\{{}\begin{matrix}\%V_{CH_4}=\dfrac{0,1}{0,15}.100\%\approx66,67\%\\\%V_{C_2H_4}\approx33,33\%\end{matrix}\right.\)
b, PT: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
Có: m tăng = mC2H4 = 0,05.28 = 1,4 (g)
a) \(n_{hh}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\)
Gọi \(\left\{{}\begin{matrix}n_{CH_4}=a\left(mol\right)\\n_{C_2H_4}=b\left(mol\right)\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}a+b=0,15\\16a+28b=3\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,1\\b=0,05\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%V_{CH_4}=\dfrac{0,1}{0,15}.100\%=66,67\%\\\%V_{C_2H_4}=100\%-66,67\%=33,33\%\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{CH_4}=\dfrac{0,1.16}{3}.100\%=53,33\%\\\%m_{C_2H_4}=100\%-53,33\%=46,67\%\end{matrix}\right.\)
b) \(m=m_{C_2H_4}=0,05.28=1,4\left(g\right)\)

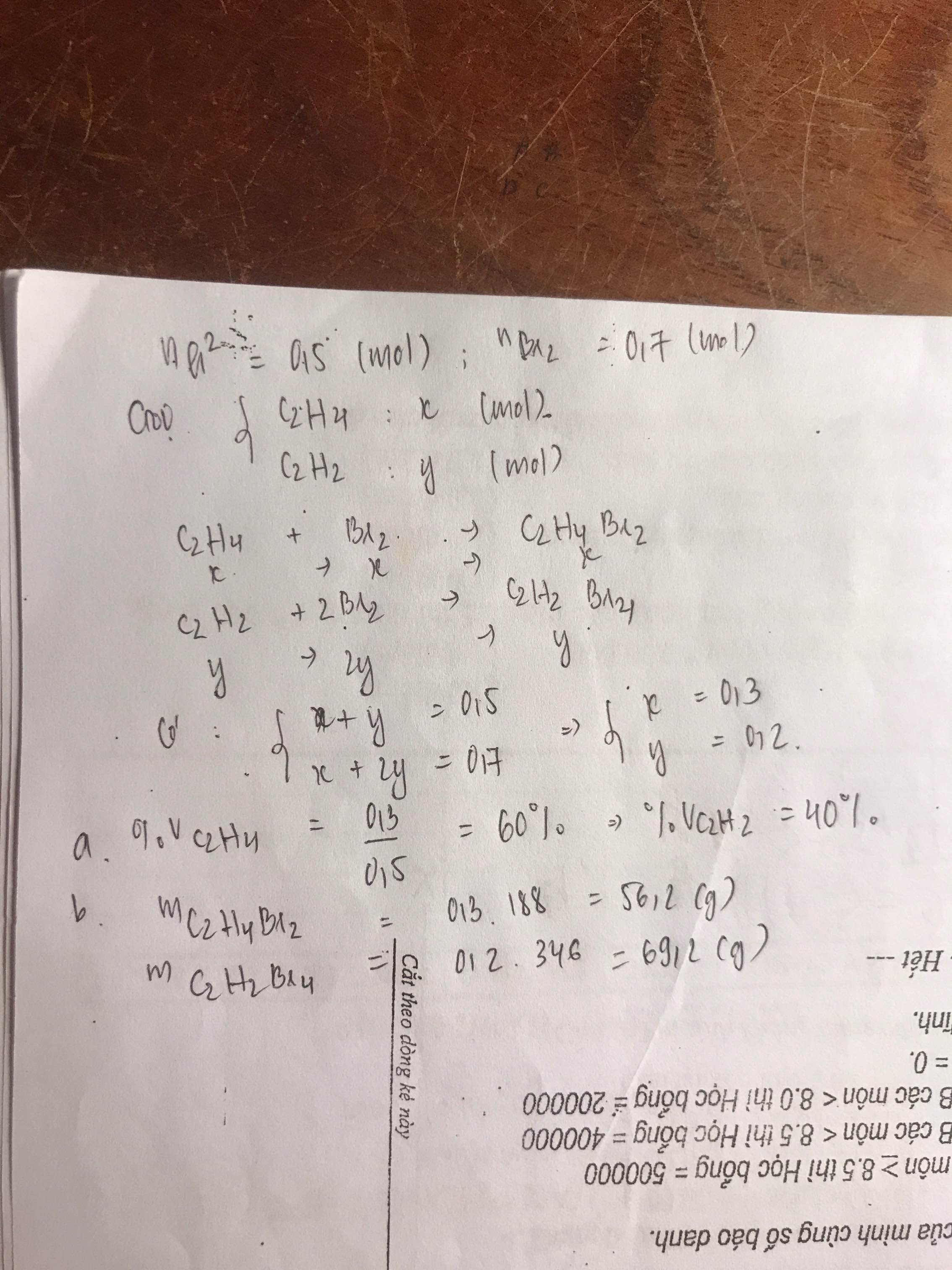
a, \(n_{C_2H_4}+n_{C_2H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\left(1\right)\)
PT: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
\(C_2H_2+2Br_2\rightarrow C_2H_2Br_4\)
Theo PT: \(n_{Br_2}=n_{C_2H_4}+2n_{C_2H_2}=\dfrac{72}{160}=0,45\left(mol\right)\left(2\right)\)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}n_{C_2H_4}=0,15\left(mol\right)\\n_{C_2H_2}=0,15\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{C_2H_4}=\dfrac{0,15.28}{0,15.28+0,15.26}.100\%\approx51,85\%\\\%m_{C_2H_2}\approx48,15\%\end{matrix}\right.\)
\(\%V_{C_2H_4}=\%V_{C_2H_2}=\dfrac{0,15.22,4}{6,72}.100\%=50\%\)
b, Theo PT: \(\left\{{}\begin{matrix}n_{C_2H_4Br_2}=n_{C_2H_4}=0,15\left(mol\right)\\n_{C_2H_2Br_4}=n_{C_2H_2}=0,15\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}m_{C_2H_4Br_2}=0,15.188=28,2\left(g\right)\\m_{C_2H_2Br_4}=0,15.346=51,9\left(g\right)\end{matrix}\right.\)