Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

goi x la so mol cua Cu
y la so mol cua CuO
\(m_{H_2SO_4}=\dfrac{70.112}{100}=78,4g\)
\(n_{H_2SO_4}=\dfrac{78,4}{98}=0,8\left(mol\right)\)
Cu+2H2SO4(d,n)\(\underrightarrow{t^o}\)CuSO2+2H2O+SO2
de: x 2x x 2x x
CuO + H2SO4\(\rightarrow\) CuSO4 +H2O
de: y y y y
Ta co: 64x + 80y = 28
2x + y = 0,8
\(\Rightarrow\left\{{}\begin{matrix}x=0,375\left(mol\right)\\y=0,05\left(mol\right)\end{matrix}\right.\)
\(m_{Cu}=0,375.64=24g\)
\(m_{CuO}=0,05.80=4g\)
\(\%m_{Cu}=\dfrac{24}{28}.100\%\approx85,71\%\)
\(\%m_{CuO}=\dfrac{4}{28}.100\%\approx14,29\%\)
mk chỉ lam ý thu 2 thoi con ý 1 mk k hieu bn muon tinh cai j

\(CuO+H_2SO_{4\left(24,5\%\right)}\rightarrow CuSO_4+H_2O\)
\(Cu+2H_2SO_{4đ}\underrightarrow{t^o}CuSO_4+SO_2+2H_2O\)
\(n_{SO_2}=\dfrac{1,12}{22,4}=0,05\left(mol\right)\)
\(\Rightarrow n_{Cu}=0,05\left(mol\right)\)
\(\Rightarrow m_{CuO}=10-64.0,05=6,8\left(g\right)\)
\(\Rightarrow n_{CuO}=0,085\left(mol\right)\)
\(\Rightarrow n_{H_2SO_4\left(24,5\%\right)}=0,085\left(mol\right)\)
\(\Rightarrow m_{H_2SO_4\left(24,5\%\right)}=8,33\left(g\right)\)
\(\Rightarrow m_{ddH_2SO_4\left(24,5\%\right)}=34\left(g\right)\)

a) \(2Fe\left(OH\right)_3-^{t^o}\rightarrow Fe_2O_3+3H_2O\)
\(Cu\left(OH\right)_2-^{t^o}\rightarrow CuO+H_2O\)
Gọi x,y lần lượt là số mol Fe(OH)3 và Cu(OH)2
=> \(\left\{{}\begin{matrix}107x+98y=20,5\\160.\dfrac{x}{2}+80y=16\end{matrix}\right.\)
=> x= 0,1 ; y=0,1
=> \(\%m_{Fe\left(OH\right)_3}=\dfrac{0,1.107}{20,5}.100=52,2\%\)
\(\%m_{Cu\left(OH\right)_2}=47,8\%\)
b) \(2Fe\left(OH\right)_3+3H_2SO_4\rightarrow Fe_2\left(SO_4\right)_3+6H_2O\)
\(Cu\left(OH\right)_2+H_2SO_4\rightarrow CuSO_4+2H_2O\)
\(n_{H_2SO_4}=0,1.\dfrac{3}{2}+0,1=0,25\left(mol\right)\)
\(m_{ddH_2SO_4}=\dfrac{0,25.98}{20\%}=122,5\left(g\right)\)
\(m_{ddsaupu}=20,5+122,5=143\left(g\right)\)
\(C\%_{Fe_2\left(SO_4\right)_3}=\dfrac{0,05.400}{143}.100=13,97\%\)
\(C\%_{CuSO_4}=\dfrac{0,1.160}{143}.100=11,19\%\)
c) \(Fe_2O_3+3H_2SO_4\rightarrow Fe_2\left(SO_4\right)_3+3H_2O\)
\(CuO+H_2SO_4\rightarrow CuSO_4+H_2O\)
\(n_{Fe_2O_3}=0,05\left(mol\right);n_{CuO}=0,1\left(mol\right)\)
=> \(n_{H_2SO_4}=0,05.3+0,1=0,25\left(mol\right)\)
\(m_{ddH_2SO_4\left(pứ\right)}=\dfrac{0,25.98}{20\%}=122,5\left(g\right)\)
=> \(m_{ddH_2SO_4\left(bđ\right)}=122,5.110\%=134,75\left(g\right)\)

PTHH: \(Mg+H_2SO_4\rightarrow MgSO_4+H_2\uparrow\)
a_______a________a______a (mol)
\(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\uparrow\)
b_______\(\dfrac{3}{2}\)b_________\(\dfrac{1}{2}\)b_____\(\dfrac{3}{2}\)b (mol)
a) Ta lập HPT: \(\left\{{}\begin{matrix}24a+27b=8,25\\a+\dfrac{3}{2}b=\dfrac{2,24}{22,4}=0,1\end{matrix}\right.\) \(\Leftrightarrow\) Hệ có nghiệm âm
*Bạn xem lại đề !!!
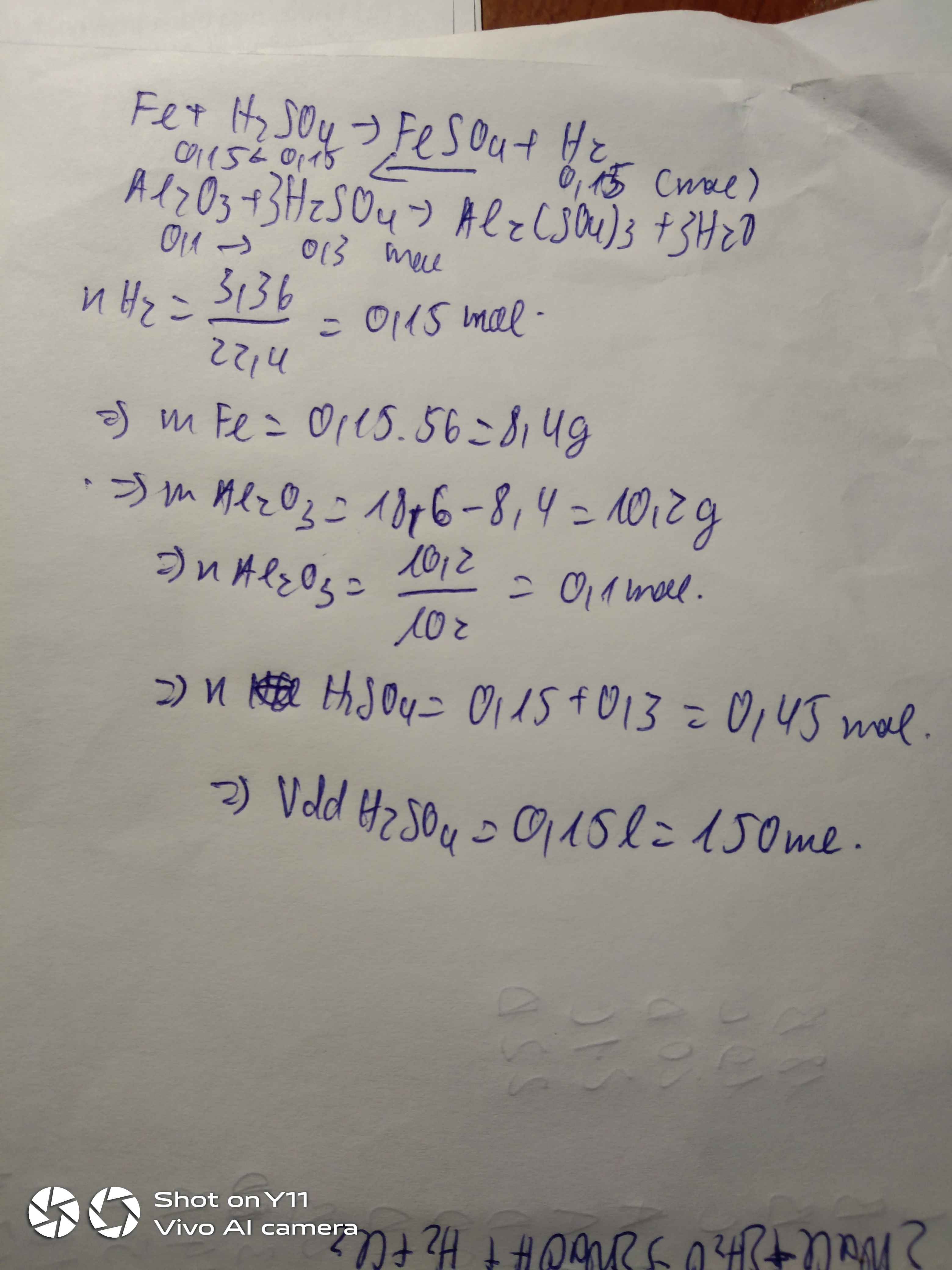

nSO2=0.25(mol)
Cu+2H2SO4->CuSo4+SO2+2H2O
CuO+H2SO4->CuSO4+H2O
nCu=nSO2=0.25(mol)
mCu=16(g)
->mCuO=12(g)
nCuO=0.15(mol)
mH2SO4=78.4
nH2SO4=0.8(mol)
tổng nH2SO4 phản ứng:0.5+0.15=0.65(mol)
nH2SO4 dư=0.15(mol)
mH2SO4 dư=14.7(g)
nCuSO4=0.4(mol)
mCuSO4=64(g)
mdd=28+112-64*0.25=124(g)
C%(H2SO4)=14.7:124*100=11.9%
C%(CuSO4)=64:124*100=51.6%
BÀI 2
mdd axit=900(g)
mH2SO4=220.5(g)
gọi mSO2 là x(g)
ta có m chất tan sau khi hòa tan=x+220.5
mdd sau khi hòa tan=x+900
theo bài ra:(x+220.5):(x+900)=49/100
100x+22050=49x+44100
51x=22050
->x=432.4(g)