Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

Ở đkc, 1 mol khí chiếm thể tích 24,79 lít
\(n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\\ PTHH:Fe+2HCl\rightarrow FeCl_2+H_2\\ \Rightarrow n_{H_2}=n_{Fe}=0,2\left(mol\right)\\ \Rightarrow V_{H_2\left(đkc\right)}=24,79\cdot0,2=4,958\left(l\right)\)

a) \(n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
PTHH: Fe + 2HCl → FeCl2 + H2
Mol: 0,2 0,4 0,2 0,2
b) \(V_{H_2}=0,2.22,4=4,48\left(l\right)\)
c) \(C\%_{ddHCl}=\dfrac{0,4.36,5.100\%}{300}=4,87\%\)
d) mdd sau pứ = 11,2 + 300 - 0,2.2 = 310,8 (g)
\(C\%_{ddFeCl_2}=\dfrac{0,2.127.100\%}{310,5}=8,17\%\)

a) PTHH: \(Zn+2HCl\rightarrow ZnCl_2+H_2\uparrow\)
\(ZnO+2HCl\rightarrow ZnCl_2+H_2O\)
b) Ta có: \(n_{H_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)=n_{Zn}\) \(\Rightarrow n_{ZnO}=\dfrac{20-0,1\cdot65}{81}=\dfrac{1}{6}\left(mol\right)\)
\(\Rightarrow n_{ZnCl_2}=n_{Zn}+n_{ZnO}=\dfrac{4}{15}\left(mol\right)\)
Mặt khác: \(m_{H_2}=0,1\cdot2=0,2\left(g\right)\)
\(\Rightarrow m_{dd}=m_{hh}+m_{ddHCl}-m_{H_2}=119,8\left(g\right)\) \(\Rightarrow C\%_{ZnCl_2}=\dfrac{\dfrac{4}{15}\cdot136}{119,8}\cdot100\%\approx30,27\%\)
c) Giả sử khí là SO2
PTHH: \(Zn+H_2SO_{4\left(đ\right)}\xrightarrow[]{t^o}ZnSO_4+SO_2\uparrow+H_2O\)
Theo PTHH: \(n_{SO_2}=n_{Zn}=0,1\left(mol\right)\) \(\Rightarrow V_{SO_2}=0,1\cdot22,4=2,24\left(l\right)\)

PTHH: \(Fe+2HCl\rightarrow FeCl_2+H_2\uparrow\)
Ta có: \(n_{HCl}=\dfrac{200\cdot71\%}{36,5}=\dfrac{284}{73}\left(mol\right)\)
\(\Rightarrow n_{Fe}=n_{FeCl_2}=n_{H_2}=\dfrac{142}{73}\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}m_{Fe}=\dfrac{142}{73}\cdot56\approx108,93\left(g\right)\\m_{FeCl_2}=\dfrac{142}{73}\cdot127\approx247,04\left(g\right)\\m_{H_2}=\dfrac{142}{73}\cdot2\approx3,89\left(g\right)\\V_{H_2}=\dfrac{142}{73}\cdot22,4\approx43,57\left(l\right)\end{matrix}\right.\)
Mặt khác: \(m_{dd}=m_{Fe}+m_{ddHCl}-m_{H_2}=305,04\left(g\right)\)
\(\Rightarrow C\%_{FeCl_2}=\dfrac{247,04}{305,04}\cdot100\%\approx80,99\%\)
Fe + 2HCl ➝ FeCl2 + H2
mHCl = 200.71% = 142 (g) => nHCl = \(\dfrac{284}{73}\) (mol)
nFe = \(\dfrac{1}{2}\) nHCl = \(\dfrac{142}{73}\) (mol) => m ≃ 108,9 (g)
nH2 = nFe => V ≃ 43,57 (l)
nFeCl2 = nFe => C% ≃ 80%
(Mk nghĩ bạn nên kiểm tra lại đề vì số liệu không được đẹp cho lắm)

Ta có: \(n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
PT: \(Fe+2HCl\rightarrow FeCl_2+H_2\)
a, Theo PT: \(n_{H_2}=n_{Fe}=0,2\left(mol\right)\Rightarrow V_{H_2}=0,2.22,4=4,48\left(l\right)\)
b, \(n_{HCl}=2n_{Fe}=0,4\left(mol\right)\Rightarrow C\%_{HCl}=\dfrac{0,4.36,5}{200}.100\%=7,3\%\)
c, \(n_{FeCl_2}=n_{Fe}=0,2\left(mol\right)\Rightarrow m_{FeCl_2}=0,2.127=25,4\left(g\right)\)
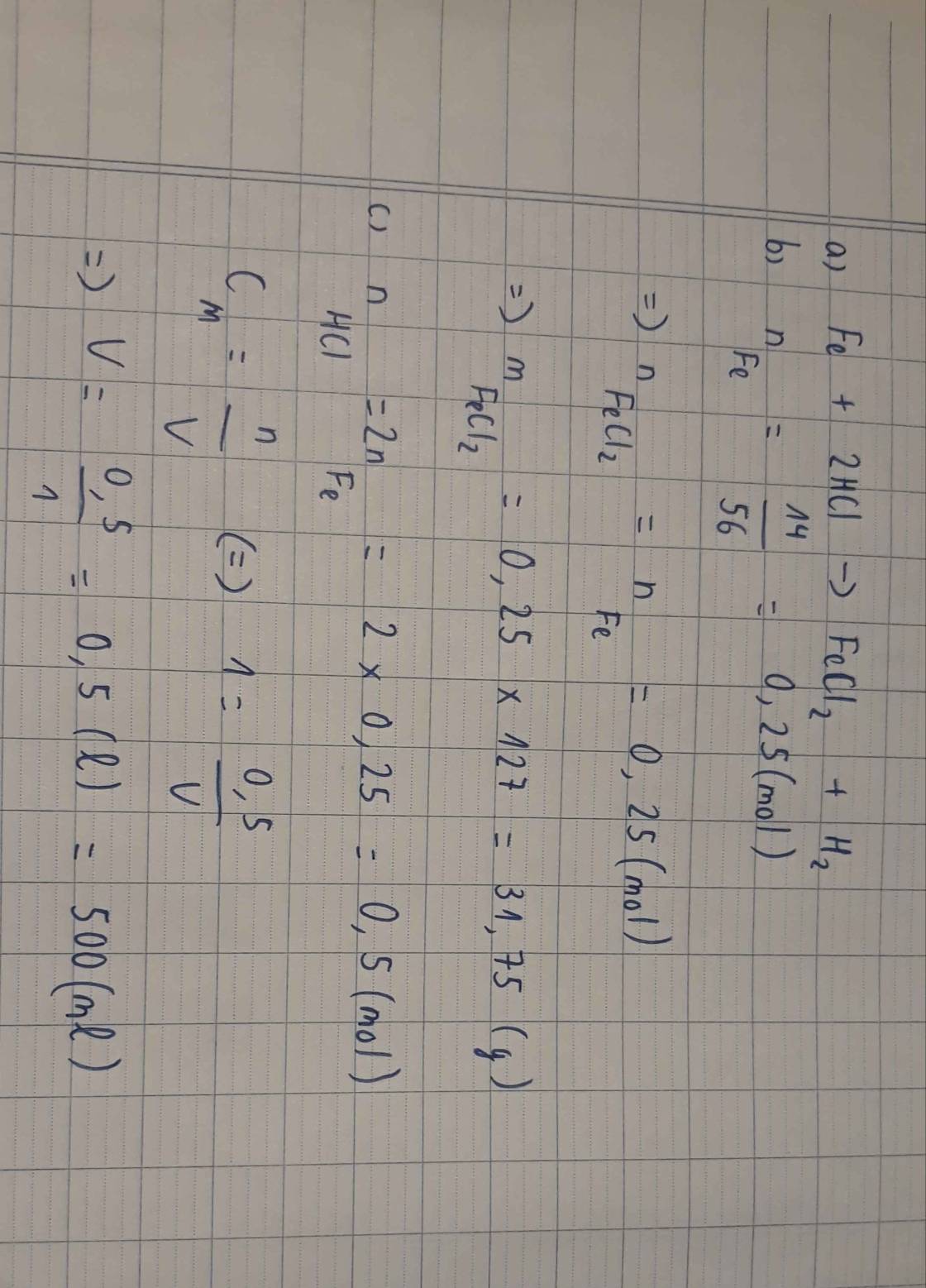
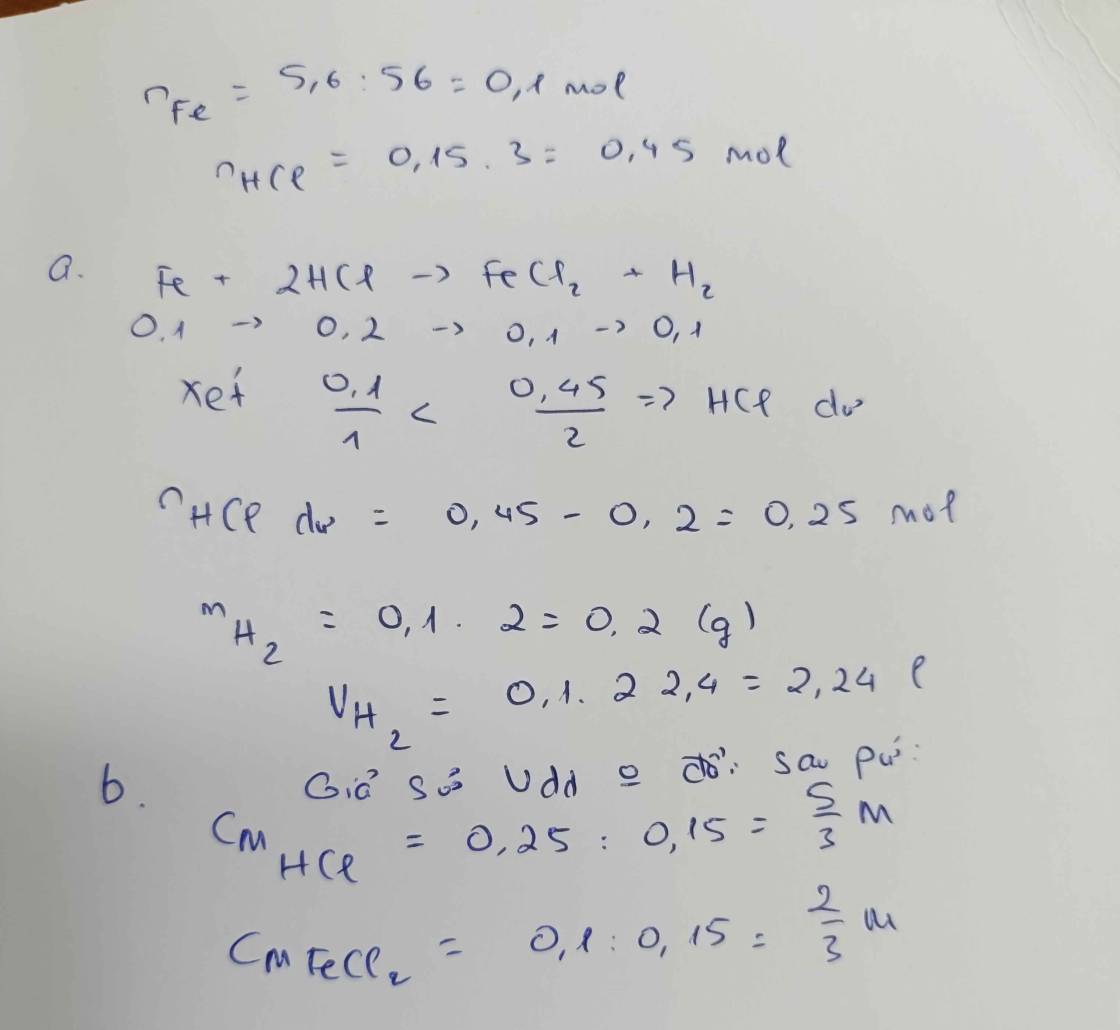

\(n_{Fe}=\dfrac{11.2}{56}=0.2\left(mol\right)\)
a: \(Fe+2HCl\rightarrow FeCl_2+H_2\)
0,2 0,2
b: V=0,2*22,4=4,48(lít)