Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a)
\(n_{Na_2CO_3}=\dfrac{10,6}{106}=0,1\left(mol\right)\)
PTHH: Na2CO3 + 2CH3COOH --> 2CH3COONa + CO2 + H2O
0,1--------->0,2------------->0,2------------>0,1
=> mCH3COOH = 0,2.60 = 12 (g)
\(C_{M\left(dd.CH_3COOH\right)}=\dfrac{0,2}{0,4}=0,5M\)
b) \(n_{C_2H_5OH}=\dfrac{13,8}{46}=0,3\left(mol\right)\)
PTHH: CH3COOH + C2H5OH --H2SO4,to--> CH3COOC2H5 + H2O
Xét tỉ lệ: \(\dfrac{0,2}{1}< \dfrac{0,3}{1}\) => Hiệu suất tính theo CH3COOH
\(n_{CH_3COOH\left(pư\right)}=\dfrac{0,2.80}{100}=0,16\left(mol\right)\)
PTHH: CH3COOH + C2H5OH --H2SO4,to--> CH3COOC2H5 + H2O
0,16------------------------------------->0,16
=> \(m_{CH_3COOC_2H_5}=0,16.88=14,08\left(g\right)\)

nFe = 11.2/56 = 0.2 (mol)
Fe + H2SO4 => FeSO4 + H2
0.2____0.2_______0.2___0.2
mH2SO4 = 0.2*98 = 19.6 (g)
mdd H2SO4 = 19.6*100/10 = 196 (g)
m dd sau phản ứng = 11.2 + 196 - 0.4 = 206.8 (g)
mFeSO4 = 0.2*152 = 30.4 (g)
C% FeSO4 = 30.4/206.8 * 100% = 14.7%
Vdd H2SO4 = mdd H2SO4 / D = 196 / 1.14 = 171.9 (ml)
CM FeSO4 = 0.2 / 0.1719 = 1.16 M

nFe = 11.2/56 = 0.2 (mol)
Fe + H2SO4 => FeSO4 + H2
0.2____0.2_______0.2___0.2
mH2SO4 = 0.2*98 = 19.6 (g)
mdd H2SO4 = 19.6*100/10 = 196 (g)
m dd sau phản ứng = 11.2 + 196 - 0.4 = 206.8 (g)
mFeSO4 = 0.2*152 = 30.4 (g)
C% FeSO4 = 30.4/206.8 * 100% = 14.7%
Vdd H2SO4 = mdd H2SO4 / D = 196 / 1.14 = 171.9 (ml)
CM FeSO4 = 0.2 / 0.1719 = 1.16 M

\(m_{CH_3COOH}=24\%.150=36\left(g\right)\\ \rightarrow n_{CH_3COOH}=\dfrac{36}{60}=0,6\left(mol\right)\)
PTHH: 2CH3COOH + Na2CO3 ---> 2CH3COONa + CO2 + H2O
0,6 0,3 0,6 0,3
=> VCO2 = 0,3.22,4 = 6,72 (l)
\(m_{Na_2CO_3}=0,3.31,8\left(g\right)\)
=> \(m_{ddNa_2CO_3}=\dfrac{31,8}{21,2\%}=150\left(g\right)\)
mCO2 = 0,3.44 = 13,2 (g)
\(m_{dd}=150+150-13,2=286,8\left(g\right)\)
\(m_{CH_3COONa}=0,3.82=24,6\left(g\right)\\ \rightarrow C\%_{CH_3COONa}=\dfrac{24,6}{286,8}=8,58\%\)

FeO+H2SO4------->FeSO4+ H2
nFeO=7,2/72=0,1 (mol)
---->nH2SO4=0,1 mol
----->mH2SO4=0,1.98=9,8(g)
---->mdung dich H2SO4=(9,8.100)/49=20(g)
--->Vdung dịch H2SO4=20/1,35=14,8(l)
2NaOH+H2SO4------>Na2SO4+2H2O
nNaOH=2.0,1=0,2(mol)
---->Vnaoh=0,2/1=0,2l

a) PTHH: \(Zn+H_2SO_4\rightarrow ZnSO_4+H_2\uparrow\)
b+c)
Ta có: \(n_{Zn}=\dfrac{19,5}{65}=0,3\left(mol\right)=n_{H_2SO_4}=n_{H_2}\)
\(\Rightarrow\left\{{}\begin{matrix}V_{H_2}=0,3\cdot22,4=6,72\left(l\right)\\m_{ddH_2SO_4}=\dfrac{0,3\cdot98}{30\%}=98\left(g\right)\end{matrix}\right.\)
d) PTHH: \(ZnSO_4+BaCl_2\rightarrow ZnCl_2+BaSO_4\downarrow\)
Ta có: \(n_{BaCl_2}=\dfrac{260\cdot20\%}{208}=0,25\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{0,3}{1}>\dfrac{0,25}{1}\) \(\Rightarrow\) ZnSO4 còn dư, BaCl2 phản ứng hết
\(\Rightarrow\left\{{}\begin{matrix}n_{ZnCl_2}=0,25mol=n_{BaSO_4}\\n_{ZnSO_4\left(dư\right)}=0,05mol\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}m_{ZnCl_2}=0,25\cdot136=34\left(g\right)\\m_{BaSO_4}=0,25\cdot233=58,25\left(g\right)\\m_{ZnSO_4\left(dư\right)}=0,05\cdot161=8,05\left(g\right)\end{matrix}\right.\)
Mặt khác: \(m_{H_2}=0,3\cdot2=0,6\left(g\right)\)
\(\Rightarrow m_{dd}=m_{Zn}+m_{ddH_2SO_4}-m_{H_2}+m_{ddBaCl_2}-m_{BaSO_4}=318,65\left(g\right)\)
\(\Rightarrow\left\{{}\begin{matrix}C\%_{ZnCl_2}=\dfrac{34}{318,65}\cdot100\%\approx10,67\%\\C\%_{ZnSO_4\left(dư\right)}=\dfrac{8,05}{318,65}\cdot100\%\approx2,53\%\end{matrix}\right.\)

nNaOH=20.10%:40=0,05 mol
2NaOH+H2SO4=Na2SO4+2H2O
=> nH2SO4=0,025 mol
=> V H2SO4=0,025/2=0,0125l=12,5 ml
Mặt khác:
2NaOH+MgCl2=Mg(OH)2+2NaCl
nNaOH=0,05 mol => nMg(OH)2=0,025 mol
=> mMg(OH)2=0,025.58=1,45g
2NaOH+H2SO4-->Na2SO4+2H2O
m NaOH=\(\frac{20.10}{100}=2\left(g\right)\)
n NaOH=\(\frac{2}{98}=0,02\left(mol\right)\)
Theo pthh
n H2SO4=1/2n NaOH=0,01(mol)
VH2SO4=\(\frac{0,01}{2}=0,005\left(mol\right)\)
2NaOH+MgCl2--->Mg(OH)2+2NaCl
Theo pthh
n Mg(OH)2=1/2 n NaOH=0,01(mol)
m Mg(OH)2=0,01.58=0,58(g)

![]()
Đặt x, y lần lượt là số mol của Na 2 CO 3 và K 2 CO 3 (x, y > 0)
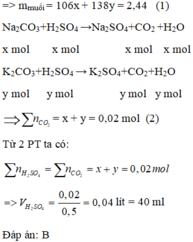

\(n_{H_2SO_4}=\dfrac{200\cdot19.6\%}{98}=0.4\left(mol\right)\)
\(SO_3+H_2O\rightarrow H_2SO_4\)
\(0.4......................0.4\)
\(m_{SO_3}=0.4\cdot80=32\left(g\right)\)
\(b.\)
\(n_{H_2SO_4}=\dfrac{80\cdot19.6\%}{98}=0.16\left(mol\right)\)
\(MgO+H_2SO_4\rightarrow MgSO_4+H_2O\)
\(0.16..........0.16..............0.16\)
\(m_{MgO}=0.16\cdot40=6.4\left(g\right)\)
\(m_{\text{dung dịch sau phản ứng}}=6.4+80=86.4\left(g\right)\)
\(C\%MgSO_4=\dfrac{0.16\cdot120}{86.4}\cdot100\%=22.22\%\)
a)
$SO_3 + H_2O \to H_2SO_4$
n SO3 = n H2SO4 = 200.19,6%/98 = 0,4(mol)
=> m = 0,4.80 = 32(gam)
b)
$MgO + H_2SO_4 \to MgSO_4 + H_2O$
n MgSO4 = n MgO = n H2SO4 = 80.19,6%/98 = 0,16(mol)
=> m MgO = 0,16.40 = 6,4(gam)
Sau pư, m dd = 6,4 + 80 = 86,4(gam)
=> C% MgSO4 = 0,16.120/86,4 .100% = 22,22%
$Na_2CO_3 + H_2SO_4 \to Na_2SO_4 + CO_2 + H_2O$
Theo PTHH :
$n_{H_2SO_4} = n_{Na_2CO_3} = \dfrac{10,6}{106} = 0,1(mol)$
$m_{dd\ H_2SO_4} = \dfrac{0,1.98}{10\%} = 98(gam)$
$V_{dd\ H_2SO_4} = \dfrac{98}{1,83} = 53,6(ml)$