Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) \(m_{HCl}=\dfrac{200.10,95}{100}=21,9\left(g\right)\)
=> \(n_{HCl}=\dfrac{21,9}{36,5}=0,6\left(mol\right)\)
b) \(n_{CaCO_3}=\dfrac{a}{100}=0,01a\left(g\right)\)
\(n_{NaOH}=0,05.2=0,1\left(mol\right)\)
PTHH: CaCO3 + 2HCl --> CaCl2 + CO2 + H2O
______0,01a---->0,02a---->0,01a->0,01a___________(mol)
NaOH + HCl --> NaCl + H2O
_0,1----->0,1___________________________________(mol)
=> 0,02a = 0,6 - 0,1
=> a = 25 (g)
c) \(V_{CO_2}=0,01.25.22,4=5,6\left(l\right)\)
d) \(\left\{{}\begin{matrix}C\%\left(CaCl_2\right)=\dfrac{0,25.111}{25+200-0,25.44}.100\%=12,97\%\\C\%\left(HCl_{dư}\right)=\dfrac{0,1.36,5}{25+200-0,25.44}.100\%=1,705\%\end{matrix}\right.\)

a) \(PT:CaCO_3+2HCl\rightarrow CaCl_2+H_2O+CO_2\uparrow\)
\(HCl+NaOH\rightarrow NaOH+H_2O\)
b) \(m_{HCl}=\frac{200.10,95\%}{100\%}=21,9\left(g\right)\)
\(n_{HCl}=\frac{21,9}{36,5}=0,6\left(mol\right)\)
c) \(n_{NaOH}=2.0,05=0,1\left(mol\right)\Rightarrow n_{HCl\left(pưNaOH\right)}=0,1\left(mol\right)\)
\(\Rightarrow n_{HCl\left(pưCaCO_3\right)}=0,6-0,1=0,5\left(mol\right)\)
d) \(n_{CaCO_3}=\frac{1}{2}n_{HCl\left(pưCaCO_3\right)}=0,5.\frac{1}{2}=0,25\left(mol\right)\)
\(m_{CaCO_3}=0,25.100=25\left(g\right)\)
e) \(n_{CO_2}=n_{CaCO_3}=0,25\left(mol\right)\)
\(V_{CO_2}=0,25.22,4=5,6\left(l\right)\)
f) \(n_{CaCl_2}=n_{CaCO_3}=0,25\left(mol\right)\)
\(m_{ddA}=25+200-0,25.44=214\left(g\right)\)
\(C\%_{ddCaCl_2}=\frac{0,25.111}{214}.100\%=12,97\%\)
\(C\%_{ddHCldư}=\frac{0,1.36,5}{214}.100\%=1,71\%\)

a, \(2Na+2H_2O\rightarrow2NaOH+H_2\)
Ta có: \(n_{Na}=\dfrac{2,3}{23}=0,1\left(mol\right)\)
\(n_{H_2O}=\dfrac{200}{18}=\dfrac{100}{9}\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{0,1}{2}< \dfrac{\dfrac{100}{9}}{2}\), ta được H2O dư.
Theo PT: \(n_{H_2}=\dfrac{1}{2}n_{Na}=0,05\left(mol\right)\Rightarrow V_{H_2}=0,05.22,4=1,12\left(l\right)\)
b, Theo PT: \(n_{NaOH}=n_{Na}=0,1\left(mol\right)\)
Ta có: m dd sau pư = 2,3 + 100 - 0,05.2 = 102,2 (g)
\(\Rightarrow C\%_{NaOH}=\dfrac{0,1.40}{102,2}.100\%\approx3,91\%\)
c, - Dung dịch làm quỳ tím hóa xanh.
\(n_{Na}=\dfrac{2,3}{23}=0,1\left(mol\right)\\ 2Na+2H_2O\rightarrow2NaOH+H_2\\ n_{H_2}=\dfrac{0,1}{2}=0,05\left(mol\right);n_{NaOH}=n_{Na}=0,1\left(mol\right)\\ a,V=V_{H_2\left(đktc\right)}=0,05.22,4=1,12\left(l\right)\\ b,m_{ddNaOH}=m_{Na}+m_{H_2O}-m_{H_2}=2,3+200-0,05.2=202,2\left(g\right)\\ C\%_{ddNaOH}=\dfrac{40.0,1}{202,2}.100\approx1,978\%\\ c,NaOH-Tính.bazo\Rightarrow Quỳ.tím.hoá.xanh\)

a, PT: \(Fe+2HCl\rightarrow FeCl_2+H_2\)
Ta có: \(n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
\(n_{HCl}=0,5.1=0,5\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{0,2}{1}< \dfrac{0,5}{2}\), ta được HCl dư.
Theo PT: \(\left\{{}\begin{matrix}n_{FeCl_2}=n_{H_2}=n_{Fe}=0,2\left(mol\right)\\n_{HCl\left(pư\right)}=2n_{Fe}=0,4\left(mol\right)\Rightarrow n_{HCl\left(dư\right)}=0,1\left(mol\right)\end{matrix}\right.\)
b, Dung dịch a gồm HCl dư và FeCl2.
PT: \(HCl+NaOH\rightarrow NaCl+H_2O\)
\(FeCl_2+2NaOH\rightarrow Fe\left(OH\right)_{2\downarrow}+2NaCl\)
Theo PT: \(n_{NaOH}=n_{HCl\left(dư\right)}+2n_{FeCl_2}=0,5\left(mol\right)\)
\(\Rightarrow a=C_{M_{NaOH}}=\dfrac{0,5}{0,2}=2,5M\)
c, \(V_{H_2}=0,2.22,4=4,48\left(l\right)\)
Bạn tham khảo nhé!

\(n_{Na}=\dfrac{6,9}{23}=0,3\left(mol\right)\\ 2Na+2H_2O\rightarrow2NaOH+H_2\\ n_{H_2}=\dfrac{0,3}{2}=0,15\left(mol\right)\\ a,V_{H_2\left(đktc\right)}=0,15.22,4=3,36\left(l\right)\\ b,m_{ddsaup.ứ}=m_{Na}+m_{H_2O}-m_{H_2}=6,9+100-0,15.2=106,6\left(g\right)\)

a, PT: \(Zn+2HCl\rightarrow ZnCl_2+H_2\)
\(Al_2O_3+6HCl\rightarrow2AlCl_3+3H_2O\)
b, Ta có: \(n_{H_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\)
Theo PT: \(n_{Zn}=n_{H_2}=0,1\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{Zn}=\dfrac{0,1.65}{20}.100\%=32,5\%\\\%m_{Al_2O_3}=67,5\%\end{matrix}\right.\)
c, Ta có: mAl2O3 = 20 - 0,1.65 = 13,5 (g)
\(\Rightarrow n_{Al_2O_3}=\dfrac{13,5}{102}=\dfrac{9}{68}\left(mol\right)\)
Theo PT: \(n_{HCl}=2n_{Zn}+6n_{Al_2O_3}=\dfrac{169}{170}\left(mol\right)\)
\(\Rightarrow V_{ddHCl}=\dfrac{\dfrac{169}{170}}{1}\approx0,994\left(l\right)\)
d, Theo PT: \(\left\{{}\begin{matrix}n_{ZnCl_2}=n_{Zn}=0,1\left(mol\right)\\n_{AlCl_3}=2n_{Al_2O_3}=\dfrac{9}{34}\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}m_{ZnCl_2}=0,1.136=13,6\left(g\right)\\m_{AlCl_3}=\dfrac{9}{34}.133,5\approx35,34\left(g\right)\end{matrix}\right.\)

a)b)c)d) mBaCl2=150.16,64%=24,96g
=>nBaCl2=0,12 mol
mH2SO4=100.14,7%=14,7g=>nH2SO4=0,15mol
BaCl2 + H2SO4 =>BaSO4 +2HCl
Bđ: 0,12 mol; 0,15 mol
Pứ: 0,12 mol=>0,12 mol=>0,12 mol=>0,24 mol
Dư: 0,03 mol
Dd ban đầu chứa BaCl2 0,12 mol và H2SO4 0,15 mol
Dd A sau phản ứng chứa HCl 0,24 mol và H2SO4 dư 0,03 mol
mHCl=0,24.36,5=8,76g
mH2SO4=0,03.98=2,94g
Kết tủa B là BaSO4 0,12 mol=>mBaSO4=0,12.233=27,96g
mddA=mddBaCl2+mddH2SO4-mBaSO4
=150+100-27,96=222,04g
C%dd HCl=8,76/222,04.100%=3,945%
C% dd H2SO4=2,94/222,04.100%=1,324%
e) HCl +NaOH =>NaCl +H2O
0,24 mol=>0,24 mol
H2SO4 +2NaOH =>Na2SO4 + 2H2O
0,03 mol=>0,06 mol
TÔNG nNaOH=0,3 mol
=>V dd NaOH=0,3/2=0,15 lit

\(n_{CO2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
Pt : \(2NaOH+CO_2\rightarrow Na_2CO_3+H_2O|\)
2 1 1 1
0,4 0,2 0,2
a) \(n_{NaOH}=\dfrac{0,2.2}{1}=0,4\left(mol\right)\)
500ml = 0,5l
\(C_{M_{ddNaOH}}=\dfrac{0,4}{0,5}=0,8\left(M\right)\)
b) \(n_{Na2CO3}=\dfrac{0,2.1}{1}=0,2\left(mol\right)\)
⇒ \(m_{Na2CO3}=0,2.106=21,2\left(g\right)\)
Chúc bạn học tốt
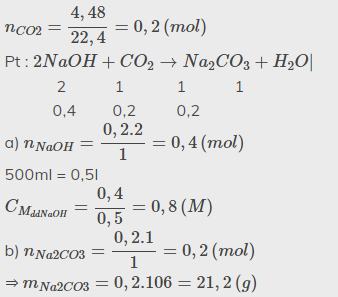
Câu 1:
\(\text{a) }pthh:CaCO3+2HCl\rightarrow CaCl_2+CO_2+H_2O\left(1\right)\\ NaOH+HCl\rightarrow NaCl+H_2O\left(2\right)\)
b) \(n_{NaOH}=C_M\cdot V=0,05\cdot2=0,1\left(mol\right)\)
Theo \(pthh\left(2\right):n_{HCl\left(2\right)}=n_{NaOH}=0,1\left(mol\right)\)
\(m_{HCl}=\dfrac{m_{d^2HCl}\cdot C\%}{100}=\dfrac{200\cdot10,95}{100}=21,9\left(g\right)\\ \Rightarrow n_{HCl}=\dfrac{m}{M}=\dfrac{21,9}{36,5}=0,6\left(mol\right)\\ \Rightarrow n_{HCl\left(1\right)}=0,6-0,1=0,5\left(mol\right)\)
Theo \(pthh\left(1\right):n_{CaCO_3}=\dfrac{1}{2}n_{HCl\left(1\right)}=\dfrac{1}{2}\cdot0,5=0,25\left(mol\right)\)
\(\Rightarrow m_{CaCO_3}=n\cdot M=0,25\cdot100=25\left(g\right)\)
c) Theo \(pthh\left(1\right):n_{CO_2}=\dfrac{1}{2}n_{HCl\left(1\right)}=\dfrac{1}{2}\cdot0,5=0,25\left(mol\right)\)
\(\Rightarrow V_{CO_2}=n\cdot22,4=0,25\cdot22,4=5,6\left(l\right)\)
d) \(m_{CO_2}=n\cdot M=0,25\cdot44=11\left(g\right)\)
\(m_{HCl\left(dư\right)}=n\cdot M=0,1\cdot36,5=3,65\left(g\right)\)
\(m_{d^2A}=\left(m_{CaCO_3}+m_{d^2HCl}\right)-m_{CO_2}\\ =\left(25+200\right)-11=214\left(g\right)\)
\(\Rightarrow C\%\left(HCl_{dư}\right)=\dfrac{3,65\cdot100}{214}=1,71\%\)
a) \(m_{NaOH}=\dfrac{m_{d^2}\cdot C\%}{100}=\dfrac{200\cdot15}{100}=30\left(g\right)\)
\(m_{d^2A\text{ sau khi pha thêm }100\left(g\right)nước}=100+200=300\left(g\right)\)
\(\Rightarrow C\%\left(NaOH\right)=\dfrac{m_{NaOH}\cdot100}{m_{d^2}}=\dfrac{30\cdot100}{300}=10\%\)
b) \(m_{NaOH\text{ sau khi cho thêm }5\left(g\right)NaOH}=30+5=35\left(g\right)\)
\(m_{d^2B}=5+200=205\left(g\right)\)
\(\Rightarrow C\%\left(NaOH\right)=\dfrac{m_{NaOH}\cdot100}{m_{d^2}}=\dfrac{35\cdot100}{205}=17,07\%\)
c) \(C\%\left(NaOH\right)=\dfrac{30\cdot100}{150}=20\%\)