Cho m gam Al tác dụng vừa đủ với V ml dung dịch axit HCl 3M, thu đc 4,2 lít khí H2. Tính m,V
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


Đặt x,y, z lần lượt là số mol của Na,Al,Mg trong m gam hỗn hợp A
m gam A + H2O dư
\(Na+H_2O\rightarrow NaOH+\dfrac{1}{2}H_2\)
x--------------------x--------->0,5x
2Al + 2NaOH + 2H2O → 2NaAlO2 + 3H2
x<------x-------------------------------------->1,5x
=> \(0,5x+1,5x=\dfrac{2,24}{22,4}=0,1\left(mol\right)\) (1)
2m gam A + NaOH
\(Na+H_2O\rightarrow NaOH+\dfrac{1}{2}H_2\)
2x------------------------------->x
2Al + 2NaOH + 2H2O → 2NaAlO2 + 3H2
2y---------------------------------------------->3y
=> \(x+3y=\dfrac{8,96}{22,4}=0,4\left(mol\right)\) (2)
3m gam A + HCl
\(Na+HCl\rightarrow NaCl+\dfrac{1}{2}H_2\)
3x--------------------------->1,5x
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
3y----------------------------->4,5y
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
3z----------------------------->3z
=> \(1,5x+4,5y+3z=\dfrac{22,4}{22,4}=1\left(mol\right)\) (3)
Từ (1), (2), (3) =>\(\left\{{}\begin{matrix}x=0,05\\y=\dfrac{7}{60}\\z=\dfrac{2}{15}\end{matrix}\right.\)
=> \(m_{Na}=0,05.23=1,15\left(g\right)\)
\(m_{Al}=\dfrac{7}{60}.27=3,15\left(g\right)\)
\(m_{Mg}=\dfrac{2}{15}.24=3,2\left(g\right)\)
=> \(m=1,15+3,15+3,2=7,5\left(g\right)\)
=> \(\%m_{Na}=\dfrac{1,15}{7,5}.100=15,33\%\)
\(\%m_{Al}=\dfrac{3,15}{7,5}.100=42\%\)
\(\%m_{Mg}=\dfrac{3,2}{7,5}.100=42,67\%\)

\(2Na+2H2O\rightarrow2NaOH+H2\left(1\right)\)
\(2Al+2NaOH+2H2O\rightarrow2NaAlO2+3H2\left(2\right)\)
\(2Al+6HCl\rightarrow2AlCl3+3H2\left(3\right)\)
\(2Na+2HCl\rightarrow2NaCl+H2\left(4\right)\)
\(Mg+2HCl\rightarrow MgCl2+H2\left(5\right)\)
\(n_{H2\left(1\right)}=0,1\left(mol\right)\rightarrow n_{Na}=0,2\left(mol\right)\rightarrow m_{Na}=4,6\left(g\right)\)
\(n_{H2\left(2\right)}=0,4\left(mol\right)\Rightarrow n_{Al}=\dfrac{4}{15}\left(mol\right)\Rightarrow m_{Al}=7,2\left(g\right)\)
\(\Rightarrow n_{H2\left(3\right)}=\dfrac{3}{2}n_{Al}=0,4\left(mol\right)\)
\(n_{H2\left(4\right)}=\dfrac{1}{2}n_{Na}=0,1\left(mol\right)\)
\(\Rightarrow n_{H2\left(5\right)}=1-0,4-0,1=0,5\left(mol\right)\)
\(\Rightarrow n_{Mg}=0,5\left(mol\right)\Rightarrow m_{Mg}=12\left(g\right)\)
\(\Rightarrow m=12+4,6+7,2=23,8\left(g\right)\)
\(\%m_{Na}=\dfrac{4,6}{23,8}.100\%=19,33\%\)
\(\%m_{Al}=\dfrac{7,2}{23,8}.100\%=30,25\%\)
\(\%m_{Mg}=100-19,33-30,25=50,42\%\)
Chúc bạn học tốt

\(\left\{{}\begin{matrix}Al\\Zn\end{matrix}\right.+HCl\rightarrow\left\{{}\begin{matrix}AlCl_3\\ZnCl_2\end{matrix}\right.+H_2\)
Bảo toàn nguyên tố H:
\(n_{HCl}=2n_{H_2}=2.\dfrac{6,72}{22,4}=0,6\left(mol\right)\)
\(\Rightarrow V=\dfrac{0,6}{2}=0,3\left(l\right)\)
\(\left\{{}\begin{matrix}AlCl_3\\ZnCl_2\end{matrix}\right.+AgNO_3\rightarrow\left\{{}\begin{matrix}Al\left(NO_3\right)_3\\Zn\left(NO_3\right)_3\end{matrix}\right.+AgCl\downarrow\)
Bào toàn nguyên tố Cl:
\(n_{AgCl}=n_{HCl}=0,6\left(mol\right)\)
\(\Rightarrow m=m_{AgCl}=0,6.143,5=86,1\left(g\right)\)

\(a.2Al+6HCl\rightarrow2AlCl_3+3H_2\\ b.n_{Al}=0,2\left(mol\right)\\ n_{HCl}=3n_{Al}=0,6\left(mol\right)\\ C\%_{HCl}=\dfrac{0,6.36,5}{150}.100=14,6\%\\ c.n_{H_2}=\dfrac{3}{2}n_{Al}=0,3\left(mol\right)\\ Bảotòannguyêntố\left(H\right)\Rightarrow n_{H_2O}=n_{H_2}=0,3\left(mol\right)\\ Bảotoànkhốilượng:m_{H_2}+m_{oxit}=m_{Fe}+m_{H_2O}\\ \Rightarrow m_{Fe}=0,3.2+17,4-0,3.18=12,6\left(g\right)\\ \Rightarrow n_{Fe}=0,225\left(mol\right)\\ Tabiết:Oxitsắtlàbaogồm:Fe,O\\ \Rightarrow m_O=17,4-12,6=4,8\left(g\right)\\ \Rightarrow n_O=0,3\left(mol\right)\\ GọiCToxitsắtlà:Fe_xO_y\left(x,y>0,x,ynguyên\right)\\ Tacó:x:y=0,225:0,3=3:4\\ VậyCToxitsắtcầntìmlàFe_3O_4\)

Gọi số mol Al, Fe là a, b (mol)
=> 27a + 56b = 11,1 (1)
\(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
a----------------------->1,5a
Fe + 2HCl --> FeCl2 + H2
b------------------------>b
=> 1,5a + b = 0,3 (2)
(1)(2) => a = 0,1; b = 0,15
=> \(\left\{{}\begin{matrix}\%m_{Al}=\dfrac{0,1.27}{11,1}.100\%=24,32\%\\\%m_{Fe}=\dfrac{0,15.56}{11,1}.100\%=75,68\end{matrix}\right.\)

nMg = 0,1(mol)
PTHH: Mg + 2HCl --> MgCl2 +H2
nMg = nMgCl2= nH2 = 0,1(mol)
=> mmuối = 9,5(g)
VH2 = 2,24(l)
b) CMHCl = 0,2/0,1=2(M)
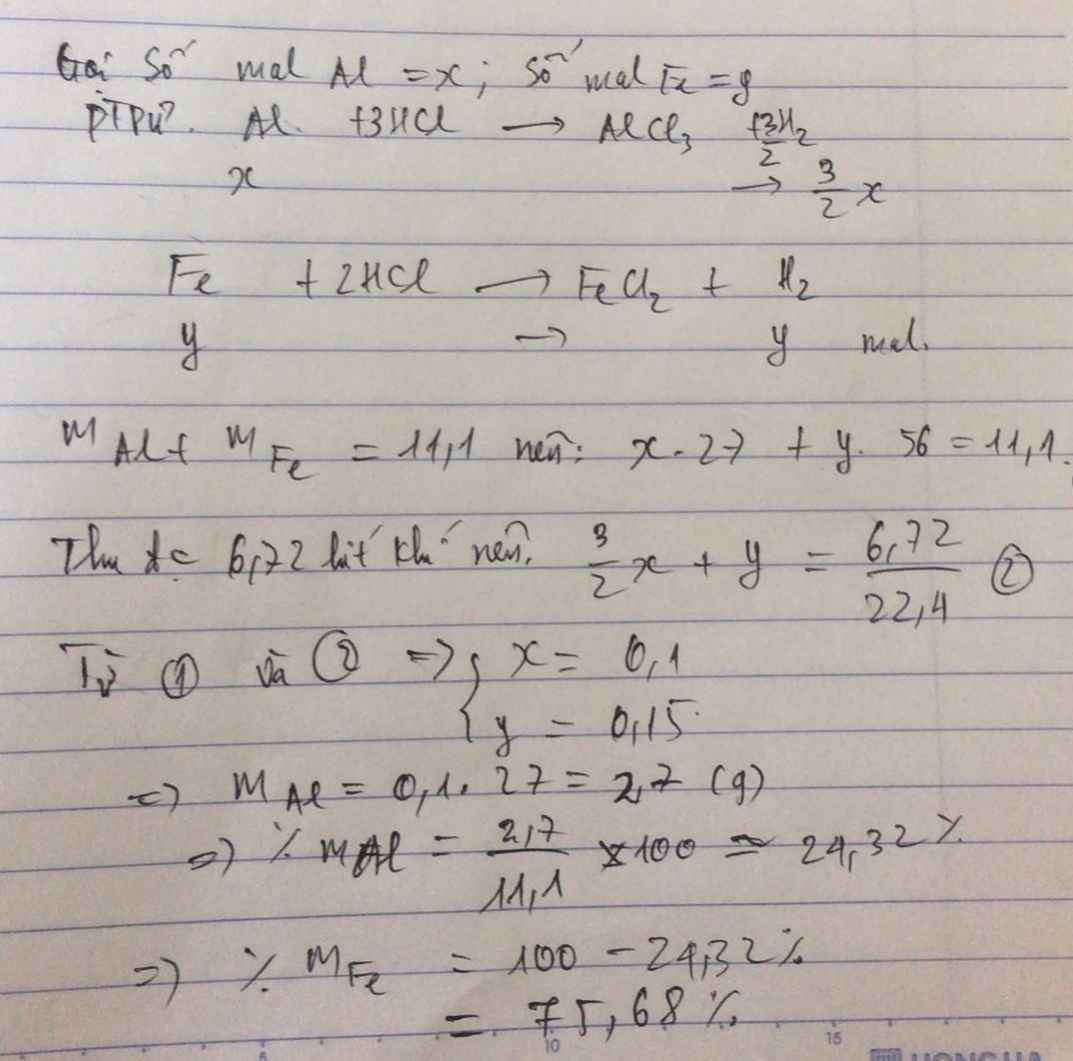
Ta có: \(n_{H_2}=\dfrac{4,2}{22,4}=0,1875\left(mol\right)\)
PT: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
__0,125__0,375___________0,1875 (mol)
\(\Rightarrow m_{Al}=0,125.27=3,375\left(g\right)\)
\(V_{HCl}=\dfrac{0,375}{3}=0,125\left(l\right)=125\left(ml\right)\)
Bạn tham khảo nhé!
n H2 = 4,2/22,4 = 3/16 mol
2Al + 6HCl $\to$ 2AlCl3 + 3H2
Theo PTHH :
n HCl = 2n H2 = 3/8 mol => V dd HCl = (3/8) / 3 = 0,125M
n Al = 2/3 n H2 = 0,125(mol) => m = 0,125.27 = 3,375(gam)