Hoàn chỉnh các phản ứng sau:
\( _{6}^{3}\textrm{Li}\) + ? → \( _{4}^{7}\textrm{Be}\) + \( _{0}^{1}\textrm{n}\).
\( _{5}^{10}\textrm{B}\) + ? → \( _{3}^{7}\textrm{Li}\) + \( _{2}^{4}\textrm{He}\)
\( _{17}^{35}\textrm{Cl}\) + ? → \( _{16}^{32}\textrm{S}\) + \( _{2}^{4}\textrm{He}\)

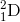 +
+ 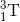 =>
=> 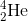 +
+ 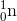 .
.
a) 3/6 li + 2/5 He-> 7/4 be + 0/1n
b)5/10B+ 0/-1 e-> 3/7li +2/4 he
c 17/35 cl + 1/1 H -> 14/32 s +2/4 he