Cho 4,11 gam kim loại bari tác dụng với 19,6 gam dung dịch axit sunfuric 10%, người ta thu được dung dịch A, kết tủa m gam B và V lít khí D (đktc). Tính nồng độ phần trăm của chất tan trong dung dịch A.
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


Gọi \(\left\{{}\begin{matrix}n_{Zn}=a\left(mol\right)\\n_{Fe}=b\left(mol\right)\end{matrix}\right.\)
\(n_{H_2}=\dfrac{15,68}{22,4}=0,7\left(mol\right)\\ m_{HCl}=200.27,375\%=54,75\left(g\right)\\ n_{HCl}=\dfrac{54,75}{36,5}=1,5\left(mol\right)\)
PTHH:
Zn + 2HCl ---> ZnCl2 + H2
a ----> 2a --------> a -----> a
Fe + 2HCl ---> FeCl2 + H2
b ---> 2b -------> b ------> b
Hệ pt \(\left\{{}\begin{matrix}65a+56b=43,7\\a+b=0,7\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,5\left(mol\right)\\b=0,2\left(mol\right)\end{matrix}\right.\)
\(\rightarrow\left\{{}\begin{matrix}m_{Zn}=0,5.65=32,5\left(g\right)\\m_{Fe}=0,2.56=11,2\left(g\right)\end{matrix}\right.\)
\(m_{dd}=43,7+200-0,7.2=242,3\left(g\right)\\ \rightarrow\left\{{}\begin{matrix}C\%_{ZnCl_2}=\dfrac{0,5.136}{242,3}=28,06\%\\C\%_{FeCl_2}=\dfrac{0,2.127}{242,3}=10,48\%\\C\%_{HCl\left(dư\right)}=\dfrac{\left(1,5-0,5.2-0,2.2\right).36,5}{242,3}=1,51\%\end{matrix}\right.\)
\(n_{H_2}=\dfrac{15,68}{22,4}=0,7\left(mol\right)\\
pthh:\left\{{}\begin{matrix}Zn+H_2SO_4->ZnSO_4+H_2\\Fe+H_2SO_4->FeSO_{\text{ 4 }}+H_2\end{matrix}\right.\)
gọi số mol Zn là x , số mol Fe là y
=> 65x+56y=43,7
=> a+b=0,7
=>a=0,5 , b =0,2
=> \(m_{Zn}=0,5.65=32,5\\ m_{Fe}=43,7-32,5=11,2\left(G\right)\)

a) \(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
\(m_{H_2SO_4}=147.10\%=14,7\left(g\right)\Rightarrow n_{H_2SO_4}=\dfrac{14,7}{98}=0,15\left(mol\right)\)
PTHH: Zn + H2SO4 → ZnSO4 + H2
Mol: 0,1 0,1 0,1 0,1
Ta có: \(\dfrac{0,1}{1}< \dfrac{0,15}{1}\) ⇒ Zn hết, H2SO4 dư
b) \(V_{H_2}=0,1.22,4=2,24\left(l\right)\)
c) mdd sau pứ = 6,5 + 147 - 0,1.2 = 153,3 (g)
\(C\%_{ddZnSO_4}=\dfrac{0,1.161.100\%}{153,3}=10,502\%\)
\(C\%_{ddH_2SO_4dư}=\dfrac{\left(0,15-0,1\right).98.100\%}{153,3}=3,196\%\)


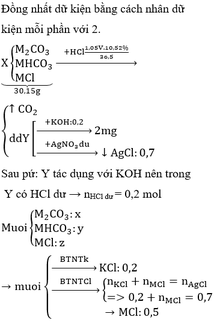
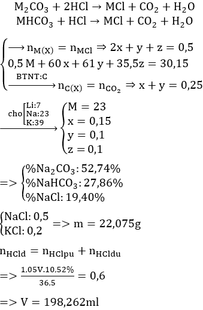
BTKL: mD + mNaHCO3 = mCO2 + mE
mD + 179,88 = 44.0,2 + 492 => mD = 320,92
BTKL: mMg + mddHCl = mH2 + mD
=> 24 . 0,4 + mddHCl = 2 . 0,4 + 320,92 => mddHCl = 312,12
=> C%HCl = 11,69%

a/
\(n_{Na_2O}=\dfrac{9,3}{62}=0,15\left(mol\right)\)
\(Na_2O+H_2O\rightarrow2NaOH\)
0,15 0,3 (mol)
\(m_{NaOH}=0,3.40=12\left(g\right)\)
\(m_A=90,7+9,3=100\left(g\right)\)
\(C\%_{NaOH}=\dfrac{12}{100}.100\%=12\%\)
b/
m\(_{FeSO_4}=\dfrac{16.200}{100}=32\left(g\right)\)
\(\rightarrow m_{FeSO_4}=\dfrac{32}{152}=\dfrac{4}{19}\left(mol\right)\)
\(2NaOH+FeSO_4\rightarrow Na_2SO_4+Fe\left(OH\right)_2\downarrow\)
bđ: 0,3 \(\dfrac{4}{19}\) 0 0 (mol)
pư: 0,3 0,15 0,15 0,15 (mol)
dư: 0 \(\dfrac{23}{380}\) (mol)
\(m_{Fe\left(OH\right)_2}=0,15.90=13,5\left(g\right)\)
\(m_C=100+200-13,5=286,5\left(g\right)\)
\(m_{Na_2SO_4}=0,15.142=21,3\left(g\right)\)
\(\rightarrow C\%_{Na_2SO_4}=\dfrac{21,3}{286,5}.100\%\approx7,4\%\)
\(m_{FeSO_4\left(dư\right)}=\dfrac{23}{380}.152=9,2\left(g\right)\)
\(\rightarrow C\%_{FeSO_4\left(dư\right)}=\dfrac{9,2}{286,5}.100\%\approx3,2\%\)

a) 2Al (0,2) + 3H2SO4 (0,3) -----> Al2(SO4)3 + 3H2 (0,3)
b) - nH2 = 0,3 mol
- Theo PTHH: nAl = 0,2 mol
=> mAl = 5,4 gam
=> mCu = 4,6 gam
==>mhh=5,4+4,6=10 g
b Theo PTHH: nH2SO4 = 0,3 mol
=> mH2SO4 = 29,4 gam
=> mdd H2SO4 = 29,4.100\20=147gam
\(2Al+3H_2SO_4-->Al_2\left(SO_4\right)_3+3H_2\left(1\right)\)
0,2______0,3_____________0,1________0,3
\(n_{H_2}=\frac{6,72}{22,4}=0,3\left(mol\right)\)
a) => \(m=0,2.27+10=15,4\left(g\right)\)
b) \(m_{d^2H_2SO_4}=\frac{0,3.98.100}{20}=147\left(g\right)\)
c) \(Ba+2H_2O-->Ba\left(OH\right)_2+H_2\left(2\right)\)
0.35__________________0,35
\(n_{Ba}=\frac{47,95}{137}=0,35\left(mol\right)\)
\(3Ba\left(OH\right)_2+Al_2\left(SO_4\right)_3-->3BaSO_4\downarrow+2Al\left(OH\right)_3\downarrow\)
0,3___________0,1_______________0,3 _______0,2
\(2Al\left(OH\right)_3+Ba\left(OH\right)_2-->Ba\left(AlO_2\right)_2+4H_2O\left(3\right)\)
0,1__________0,05__________0,05
\(m_{d^2sau}=0,2.27+147-0,3.2+47,95-0,3.233-0,2.78=137,55\left(g\right)\)
\(C\%_{Ba\left(AlO_2\right)_2}=\frac{0,05.255}{137,55}.100=9,27\%\)

a) \(n_{Al}=\dfrac{8,64}{27}=0,32\left(mol\right)\)
\(n_{HCl}=\dfrac{365.10\%}{36,5}=1\left(mol\right)\)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
Xét tỉ lệ \(\dfrac{0,32}{2}< \dfrac{1}{6}\) => Al hết, HCl dư
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
0,32-->0,96---->0,32--->0,48
=> \(V_{H_2}=0,48.22,4=10,752\left(l\right)\)
b) Trong Y chứa AlCl3 và HCl dư
\(m_{AlCl_3}=0,32.133,5=42,72\left(g\right)\)
c) mdd sau pư = 8,64 + 365 - 0,48.2 = 372,68 (g)
\(\left\{{}\begin{matrix}C\%\left(AlCl_3\right)=\dfrac{42,72}{372,68}.100\%=11,463\%\\C\%\left(HCldư\right)=\dfrac{\left(1-0,96\right).36,5}{372,68}.100\%=0,392\%\end{matrix}\right.\)