Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

Ta có: \(n_K=\dfrac{7,8}{39}=0,2\left(mol\right)\)
a, PT: \(2K+2CH_3COOH\rightarrow2CH_3COOK+H_2\)
_____0,2______0,2_____________________0,1 (mol)
b, \(m_{CH_3COOH}=0,2.60=12\left(g\right)\)
c, \(V_{H_2}=0,1.22,4=2,24\left(l\right)\)
Bạn tham khảo nhé!

\(n_{Na}=\dfrac{9,2}{23}=0,4mol\)
\(Na+CH_3COOH\rightarrow CH_3COONa+\dfrac{1}{2}H_2\)
0,4 0,4 ( mol )
\(m_{CH_3COOH}=0,4.60=24g\)

n$Mg$ =4,8/24=0,2 mol
n$CH3COOH$ =12/60=0,2 mol
Xét tỉ lệ mol=>$CH3COOH$ hết
2 $CH3COOH$ +$Mg$ => $(CH3COO)2Mg$ + $H_2$
0,2 mol =>0,1 mol
V$H_2$ =0,1.22,4=2,24l

a, \(Na_2CO_3+2HCl\rightarrow2NaCl+CO_2+H_2O\)
b,\(n_{Na_2CO_3}=\dfrac{21,2}{106}=0,2\left(mol\right)\)
Theo PT: \(n_{HCl}=2n_{Na_2CO_3}=0,4\left(mol\right)\)
\(\Rightarrow m_{HCl}=0,4.36,5=14,6\left(g\right)\)
c, \(n_{CO_2}=n_{Na_2CO_3}=0,2\left(mol\right)\)
\(\Rightarrow V_{CO_2}=0,2.22,4=4,48\left(l\right)\)

a) PTHH: \(Zn+2HCl\rightarrow ZnCl_2+H_2\uparrow\)
b+c) Ta có: \(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}n_{HCl}=0,2\left(mol\right)\\n_{H_2}=0,1\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}m_{HCl}=0,2\cdot36,5=7,3\left(g\right)\\V_{H_2}=0,1\cdot22,4=2,24\left(l\right)\end{matrix}\right.\)

\(n_{Mg}=\dfrac{2,4}{24}=0,1\left(mol\right)\)
\(Mg+2CH_3COOH\rightarrow\left(CH_3COO\right)_2Mg+H_2\)
0,1 0,2
a. \(V_{CH_3COOH}=\dfrac{0,2}{1}=0,2\left(l\right)\)
b. \(CH_3COOH+C_2H_5OH⇌\left(H_2SO_{4đ},t^o\right)CH_3COOC_2H_5+H_2O\)
0,2 0,2
Với H% = 80
\(m_{CH_3COOC_2H_5}=\dfrac{0,2.88.80}{100}=14,08\left(g\right)\)
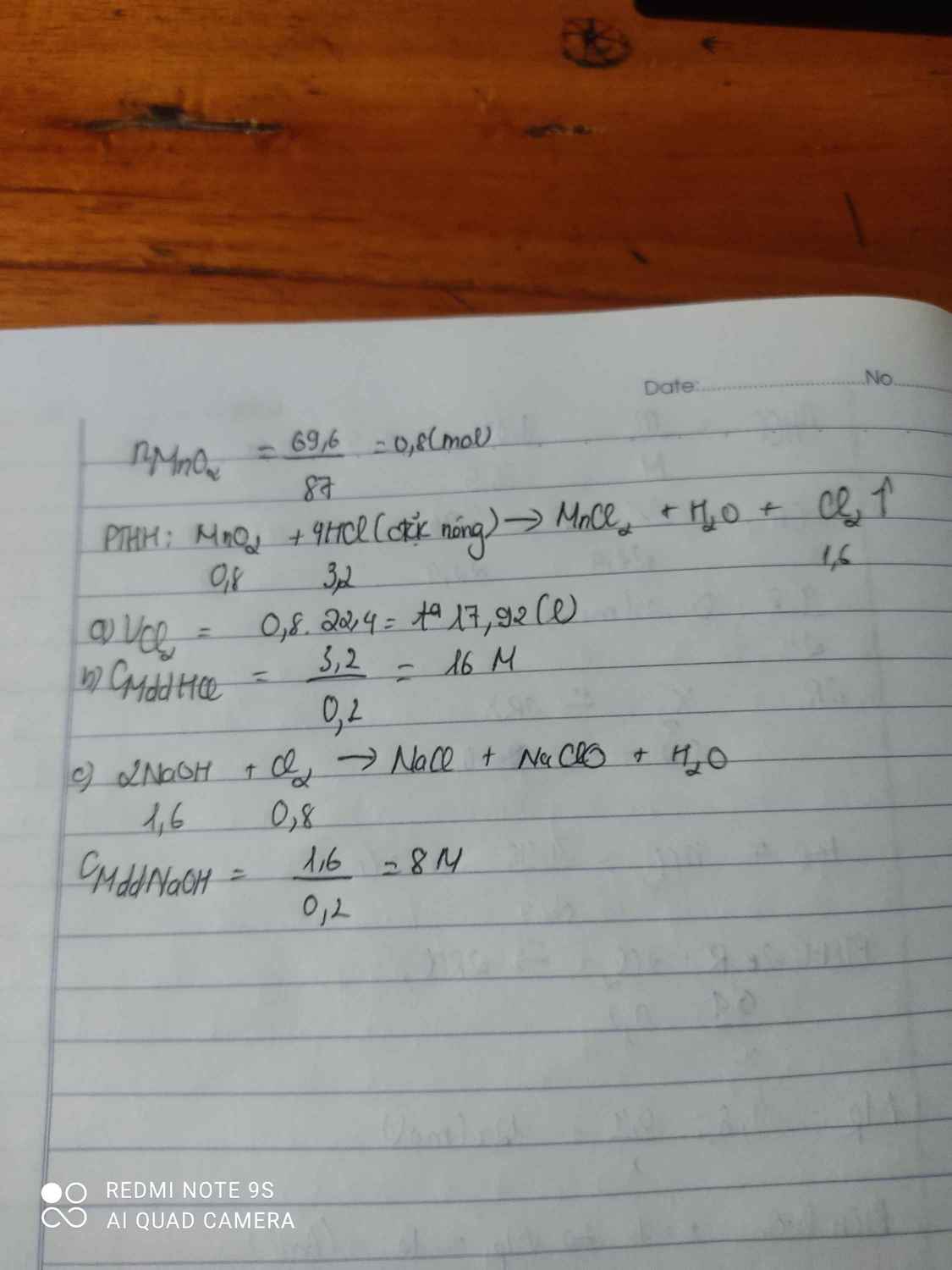
$a\big)$
$n_{Na}=\dfrac{4,6}{23}=0,2(mol)$
$CH_3COOH+Na\to CH_3COONa+\dfrac{1}{2}H_2$
Theo PT: $n_{CH_3COOH}=n_{Na}=0,2(mol)$
$\to m_{CH_3COOH}=0,2.60=12(g)$
$b\big)$
Theo PT: $n_{H_2}=\dfrac{1}{2}n_{Na}=0,1(mol)$
$\to V_{H_2(đktc)}=0,1.22,4=2,24(l)$
giúp tuiii