Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

bổ sung:
\(m_{dd}=400+0,2.56=411,2\left(g\right)\)
\(C\%_{H2SO4}dư=\dfrac{\left(0,2-0,1\right)98}{411,2}.100\%=2,38\%\)
\(n_{K2SO4}=\dfrac{1}{2}n_{KOH}=0,1\left(mol\right)\)
\(C\%_{K2SO4}=\dfrac{0,1.174}{411,2}.100\%=4,23\%\)
Chúc bạn học tốt

\(n_{H_2SO_4}=0,25.2=0,5\left(mol\right)\)
Gọi \(\left\{{}\begin{matrix}n_{Al_2O_3}=x\left(mol\right)\\n_{CuO}=y\left(mol\right)\end{matrix}\right.\)
\(Al_2O_3+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2O\)
x----------> 3x --------> x
\(CuO+H_2SO_4\rightarrow CuSO_4+H_2O\)
y --------> y --------> y
Có hệ phương trình
\(\left\{{}\begin{matrix}102x+80y=26,2\\3x+y=0,5\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}x=0,1\\y=0,2\end{matrix}\right.\)
\(\%_{m_{Al_2O_3}}=\dfrac{102.0,1.100}{26,2}=38,93\%\)
\(\%_{m_{CuO}}=\dfrac{80.0,2.100}{26,2}=61,07\%\)
\(CM_{Al_2\left(SO_4\right)_3}=\dfrac{x}{0,25}=\dfrac{0,1}{0,25}=0,4M\)
\(CM_{CuSO_4}=\dfrac{y}{0,25}=\dfrac{0,2}{0,25}=0,8M\)

\(n_{SO_3}=\dfrac{80}{80}=1mol\\ m_{H_2SO_4\left(bđ\right)}=1000\cdot1,2\cdot10\%=120g\\ SO_3+H_2O->H_2SO_4\\ C_{\%}=\dfrac{120+98}{1000\cdot1,2+80}\cdot100\%=17,03\%\)

PT: \(Zn+H_2SO_4\rightarrow ZnSO_4+H_2\)
a, Ta có: \(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
\(n_{H_2SO_4}=0,2.1=0,2\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{0,1}{1}< \dfrac{0,2}{1}\). ta được H2SO4 dư.
Theo PT: \(n_{H_2SO_4\left(pư\right)}=n_{ZnSO_4}=n_{Zn}=0,1\left(mol\right)\)
\(\Rightarrow n_{H_2SO_4\left(dư\right)}=0,2-0,1=0,1\left(mol\right)\)
b, Ta có: \(m_{ZnSO_4}=0,1.161=16,1\left(g\right)\)
c, \(C_{M_{H_2SO_4\left(dư\right)}}=\dfrac{0,1}{0,2}=0,5M\)
\(C_{M_{ZnSO_4}}=\dfrac{0,1}{0,2}=0,5M\)
Bạn tham khảo nhé!

a, \(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
b, \(n_{Fe}=\dfrac{1,12}{56}=0,02\left(mol\right)\)
\(n_{H_2SO_4}=0,2.0,12=0,024\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{0,02}{1}< \dfrac{0,024}{1}\), ta được H2SO4 dư.
Theo PT: \(n_{H_2SO_4\left(pư\right)}=n_{FeSO_4}=n_{Fe}=0,02\left(mol\right)\)
\(\Rightarrow n_{H_2SO_4\left(dư\right)}=0,024-0,02=0,004\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}C_{M_{FeSO_4}}=\dfrac{0,02}{0,2}=0,1\left(M\right)\\C_{M_{H_2SO_4\left(dư\right)}}=\dfrac{0,004}{0,2}=0,02\left(M\right)\end{matrix}\right.\)

PT: \(Al_2O_3+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2O\)
a, Ta có: \(n_{Al_2O_3}=\dfrac{10,2}{102}=0,1\left(mol\right)\)
\(m_{H_2SO_4}=\dfrac{200.20}{100}=40\left(g\right)\Rightarrow n_{H_2SO_4}=\dfrac{40}{98}=\dfrac{20}{49}\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{0,1}{1}< \dfrac{\dfrac{20}{49}}{3}\), ta được H2SO4 dư.
Theo PT: \(n_{H_2SO_4\left(pư\right)}=3n_{Al_2O_3}=0,3\left(mol\right)\)
\(\Rightarrow n_{H_2SO_4\left(dư\right)}=\dfrac{53}{490}\left(mol\right)\)
\(\Rightarrow m_{H_2SO_4\left(dư\right)}=\dfrac{53}{490}.98=10,6\left(g\right)\)
b, Theo PT: \(n_{Al_2\left(SO_4\right)_3}=n_{Al_2O_3}=0,1\left(mol\right)\)
\(\Rightarrow m_{Al_2\left(SO_4\right)_3}=0,1.342=34,2\left(g\right)\)
c, Ta có: m dd sau pư = mAl2O3 + m dd H2SO4 = 210,2 (g)
\(\Rightarrow\left\{{}\begin{matrix}C\%_{H_2SO_4\left(dư\right)}=\dfrac{10,6}{210,2}.100\%\approx5,04\%\\C\%_{Al_2\left(SO_4\right)_3}=\dfrac{34,2}{210,2}.100\%\approx16,3\%\end{matrix}\right.\)
Bạn tham khảo nhé!

300ml NaOH 0,1 M
=> n OH- = 0,1 * 0,3 = 0,03 mol
300ml Ba(OH)2 0,025 M
=> n OH- = 2 * 0,025 * 0,3 = 0,015 mol
vậy n OH- = 0,03 + 0,015 = 0,045 mol
200ml dd H2SO4 có nồng độ x M
=> n H+ = 2 * 0,2 * x = 0,4x mol
sau phản ứng thu được 500ml dd có pH=2
pH=2 => [H+] dư = 10^-2 = 0,01 M
=> n H+ dư = 0,01 * 0,5 = 0,005 mol
H+ + OH- ---> H2O
0,045 <---0,045
ta có: 0,4x = 0,045 + 0,005 (số mol đề bài = số mol pu + số mol dư)
=> x = 0,125 M
--------------------------------------...
n SO4 2- = 0,2x = 0,2 * 0,125 = 0,025 mol
n Ba 2+ = 0,025 * 0,3 = 0,0075 mol
Ba 2+ + SO4 2- ---> BaSO4
0,0075 --> 0,0075
m BaSO4 = 0,0075 * 233 = 1,7475 g
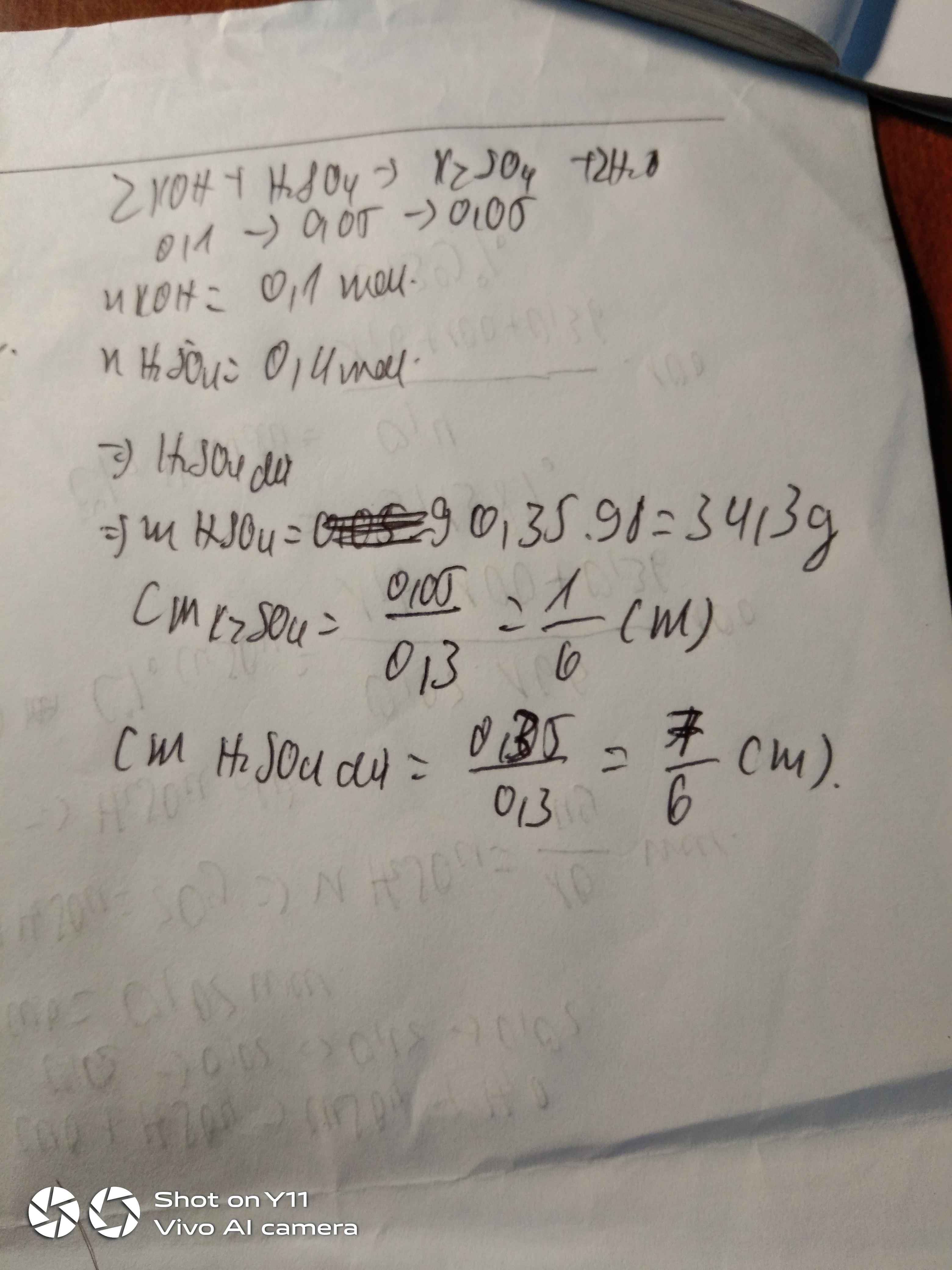
D = 1,1 g/ml mới đúng
\(n_{Al_2O_3}=\dfrac{1,02}{102}=0,01\left(mol\right)\)
\(m_{dd.H_2SO_4}=200.1,1=220\left(g\right)\)
\(n_{H_2SO_4}=\dfrac{220.4,9}{100}:98=0,11\left(mol\right)\)
\(Al_2O_3+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2O\)
0,01 ----> 0,03 ------> 0,01
Xét \(\dfrac{0,11}{1}< \dfrac{0,11}{3}\) => \(H_2SO_4\)dư
\(n_{H_2SO_4.dư}=0,11-0,03=0,08\left(mol\right)\Rightarrow CM_{H_2SO_4}=\dfrac{0,08}{0,2}=0,4M\)
\(n_{Al_2\left(SO_4\right)_3}=0,01\rightarrow CM_{Al_2\left(SO_4\right)_3}=\dfrac{0,01}{0,2}=0,05M\)
\(m_{dd.muối}=1,02+220=221,02\left(g\right)\)
\(C\%_{H_2SO_4}=\dfrac{0,08.98.100}{221,02}=3,55\%\)
\(C\%_{Al_2\left(SO_4\right)_3}=\dfrac{342.0,01.100}{221,02}=1,55\%\)