Trung hòa 20ml dd HNO3 1M(D=1.12g/ml) bằng một lượng vừa đủ NaOH 4% thu đc dd X.
a/Tính khối lượng dd NaOH đã dùng.
b/ tính C% của chất có trong dd X.
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a/ \(m_{H_2SO_4}=490.10\%=49\left(g\right)\Rightarrow n_{H_2SO_4}=\dfrac{49}{98}=0,5\left(mol\right)\)
PTHH: 2NaOH + H2SO4 → Na2SO4 + 2H2O
Mol: 1 0,5 0,5
\(m_{ddNaOH}=\dfrac{1.40.100}{20}=200\left(g\right)\)
b/ \(m_{Na_2SO_4}=0,5.142=71\left(g\right)\)

a, nH2SO4=0.02*1=0.02(mol)
H2SO4 + NaOH ➞ Na2SO4 +H2O
0.02.........0.02........0.02.........0.02.......(mol)
m dung dịch NaOH=(0.02*40)*100/20=4(g)
b) H2SO4 + KOH ➞ K2SO4 +H2O
....0.02.......0.02..........0.02......0.02...(mol)
mdung dịch KOH=(0.02*56)*100/5.6=20(g)
Vdung dịch=20/1.045=19.139(ml)

a) H2SO4 + 2NaOH --> Na2SO4 + 2H2O
b) \(n_{H_2SO_4}=0,2.1=0,2\left(mol\right)\)
PTHH: H2SO4 + 2NaOH --> Na2SO4 + 2H2O
0,2---->0,4
=> mNaOH = 0,4.40 = 16 (g)
=> \(m_{dd.NaOH}=\dfrac{16.100}{20}=80\left(g\right)\)
c)
PTHH: H2SO4 + 2KOH --> K2SO4 + 2H2O
0,2---->0,4
=> mKOH = 0,4.56 = 22,4 (g)
=> \(m_{dd.KOH}=\dfrac{22,4.100}{5,6}=400\left(g\right)\)
=> \(V_{dd.KOH}=\dfrac{400}{1,045}=382,775\left(ml\right)\)
\(n_{H_2SO_4}=0,2.1=0,2\left(mol\right)\\ pthh:H_2SO_4+2NaOH\rightarrow Na_2SO_4+2H_2O\)
0,2 0,4
\(m_{\text{ }NaOH}=0,4.40=16g\\ m_{\text{dd}NaOH}=\dfrac{16.100}{20}=80g\)
\(pthh:H_2SO_4+2KOH\rightarrow K_2SO_4+2H_2O\)
0,2 0,4
\(m_{KOH}=0,4.56=22,4g\\
m_{\text{dd}KOH}=\dfrac{22,4.100}{5,6}=400g\\
V_{\text{dd}}=\dfrac{400}{1,045}=382,7ml\)
\(V_{dd}=\dfrac{22,4}{1,045}=21,4354ml\)

Ta có; \(\left\{{}\begin{matrix}n_{NaOH}=0,02.2=0,04\left(mol\right)\\n_{KOH}=0,01.2=0,02\left(mol\right)\end{matrix}\right.\)
PTHH: \(2NaOH+H_2SO_4\rightarrow Na_2SO_4+2H_2O\)
pư..............0,04..........0,02..............0,02............0,04 (mol)
PTHH: \(2KOH+H_2SO_4\rightarrow K_2SO_4+2H_2O\)
pư............0,02..........0,01.............0,01...........0,02 (mol)
\(\Rightarrow C_{M_{ddH2SO4}}=\dfrac{0,02+0,01}{0,03}=1\left(M\right)\)
Tương tự ta có:\(\left\{{}\begin{matrix}n_{H_2SO_4}=0,02.1=0,02\left(mol\right)\\n_{HCl}=0,005.1=0,005\left(mol\right)\end{matrix}\right.\)
PTHH: \(2NaOH+H_2SO_4\rightarrow Na_2SO_4+2H_2O\)
pư.............0,04.............0,02............0,02............0,04 (mol)
PTHH: \(NaOH+HCl\rightarrow NaCl+H_2O\)
pư............0,005.....0,005.......0,005.....0,005 (mol)
\(\Rightarrow C_{M_{ddNaOH}}=\dfrac{0,04+0,005}{0,03}=3\left(M\right)\)
Vậy......

a) $2NaOH + H_2SO_4 \to Na_2SO_4 + 2H_2O$
b)
n H2SO4 = 0,03.1 = 0,03(mol)
n NaOH = 2n H2SO4 = 0,06(mol)
=> CM NaOH = 0,06/0,05 = 1,2M
c) $H_2SO_4 + 2KOH \to K_2SO_4 + 2H_2O$
n KOH = 2n H2SO4 = 0,06(mol)
=> m KOH = 0,06.56 = 3,36 gam
=> m dd KOH = 3,36/5,6% = 60(gam)
=> V dd KOH = m/D = 60/1,045 = 57,42(ml)

\(a.Na_2CO_3+2HCl\rightarrow2NaCl+CO_2+H_2O\\ n_{NaCl}=n_{HCl}=2.n_{CO_2}=2.\dfrac{448:1000}{22,4}=0,04\left(mol\right)\\ C_{MddHCl}=\dfrac{0,04}{0,02}=2\left(M\right)\\ b.m_{NaCl}=58,5.0,04=2,34\left(g\right)\\ c.m_{Na_2CO_3}=106.0,02=2,12\left(g\right)\\ \%m_{Na_2CO_3}=\dfrac{2,12}{5}.100=42,4\%\\ \%m_{NaCl}=100\%-42,4\%=57,6\%\)
Bài 16 :
\(n_{CO2}=\dfrac{0,448}{22,4}=0,02\left(mol\right)\)
Pt : \(Na_2CO_3+2HCl\rightarrow2NaCl+CO_2+H_2O|\)
1 2 2 1 1
0,02 0,04 0,04 0,02
a) \(n_{HCl}=\dfrac{0,02.2}{1}=0,04\left(mol\right)\)
20ml = 0,02l
\(C_{M_{HCl}}=\dfrac{0,04}{0,02}=2\left(M\right)\)
b) \(n_{NaCl}=\dfrac{0,02.2}{1}=0,04\left(mol\right)\)
⇒ \(m_{NaCl}=0,04.58,5=2,34\left(g\right)\)
c) \(n_{Na2CO3}=\dfrac{0,04.1}{2}=0,02\left(mol\right)\)
⇒ \(m_{Na2CO3}=0,02.106=2,12\left(g\right)\)
\(m_{NaCl}=5-2,12=2,88\left(g\right)\)
0/0Na2CO3 = \(\dfrac{2,12.100}{5}=42,4\)0/0
0/0NaCl = \(\dfrac{2,88.100}{5}=57,6\)0/0
Chúc bạn học tốt

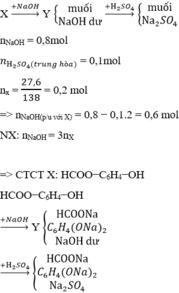
nNaOH = 0,2mol
n H 2 S O 4 ( t r u n g h ò a ) = 0,02mol
nx = 9,12/152 = 0,06 mol => nNaOH(p/u với X) = 0,2 − 0,01.2 = 0,18 mol
−NX: nNaOH = 3nX
=> CTCTX: RCOO−C6H4(R′)−OH
Với R +R' = CH3
RCOO−C6H4(R′)−OH  RCOONa + C6H4(R′)(ONa)2 + 2H2O
RCOONa + C6H4(R′)(ONa)2 + 2H2O
2NaOH +H2SO4 → Na2SO4 + 2H2O
=> nH2O = 2nX + 2 nNaOH dư = 2. 0,06 + 0,01.2 =0,14
Bảo toàn khối lượng cho cả 2 quá trình:
mX + mNaOH + mH2SO4 = mrắn + mH2O
=> mrắn = 15,58 gam
Đáp án cần chọn là: C

a, PT: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(FeO+2HCl\rightarrow FeCl_2+H_2O\)
b, Ta có: \(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
Theo PT: \(n_{Al}=\dfrac{2}{3}n_{H_2}=0,2\left(mol\right)\)
\(\Rightarrow m_{Al}=0,2.27=5,4\left(g\right)\)
⇒ mFeO = 12,6 - 5,4 = 7,2 (g)
c, Phần này đề cho dd NaOH dư hay vừa đủ bạn nhỉ?
d, Cho hh vào dd H2SO4 đặc nguội thì có khí thoát ra.
PT: \(2FeO+4H_2SO_{4\left(đ\right)}\rightarrow Fe_2\left(SO_4\right)_3+SO_2+4H_2O\)
Ta có: \(n_{FeO}=\dfrac{7,2}{72}=0,1\left(mol\right)\)
Theo PT: \(n_{SO_2}=\dfrac{1}{2}n_{FeO}=0,05\left(mol\right)\)
\(\Rightarrow V_{SO_2}=0,05.22,4=1,12\left(l\right)\)

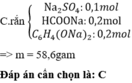
\(a/n_{HNO_3}=0,02.1=0,02mol\\ HNO_3+NaOH\rightarrow NaNO_3+H_2O\\ n_{NaOH}=n_{NaOH}=n_{NaNO_3}=0,02mol\\ m_{ddNaOH}=\dfrac{0,02.40}{4\%}\cdot100\%=20g\\ b/m_{ddHNO_3}=20.1,12=22,4g\\ C_{\%NaNO_3}=\dfrac{0,02.85}{22,4+20}\cdot100\%\approx4,01\%\)