Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) Fe + H2SO4 → FeSO4 + H2
b) Ta có : nH2 = \(\dfrac{16,8}{22,4}\) = 0,75 (mol)
⇒ nFe= 0,75.56 = 42(gam)

PT: \(Fe+H_2SO_4\rightarrow FeSO_4+H_2\) (1)
\(2Fe+6H_2SO_{4\left(đ\right)}\underrightarrow{t^o}Fe_2\left(SO_4\right)_3+3SO_2+6H_2O\) (2)
\(5SO_2+2KMnO_4+2H_2O\rightarrow2MnSO_4+K_2SO_4+2H_2SO_4\) (3)
Ta có: \(n_{H_2}=0,2\left(mol\right)\)
Theo PT (1): \(n_{Fe}=n_{H_2}=0,2\left(mol\right)\)
Theo PT (2): \(n_{SO_2}=\dfrac{3}{2}n_{Fe}=0,3\left(mol\right)\)
\(\Rightarrow V_{SO_2}=0,3.22,4=6,72\left(l\right)\)
Theo PT (3): \(n_{KMnO_4}=\dfrac{2}{5}n_{SO_2}=0,12\left(mol\right)\)
\(\Rightarrow V_{KMnO_4}=\dfrac{0,12}{2}=0,06\left(l\right)\)
Bạn tham khảo nhé!

a)
$Fe + 2HCl \to FeCl_2 + H_2$
Theo PTHH :
$n_{Fe} = n_{H_2} = \dfrac{11,2}{22,4} = 0,5(mol)$
$A = 0,5.56 = 28(gam)$
b) $n_{HCl} = 2n_{H_2} = 1(mol)$
$m_{HCl} = 1.36,5 = 36,5(gam)$
c) $m_{dd\ HCl} = 36,5 : 20\% = 182,5(gam)$
$m_{dd\ sau\ pư} = 28 + 182,5 - 0,5.2 = 209,5(gam)$
$C\%_{FeCl_2} = \dfrac{0,5.127}{209,5}.100\% = 30,3\%$

a)
\(n_{H_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\)
PTHH: Mg + 2HCl --> MgCl2 + H2
_____0,1<---0,2<-------0,1<---0,1
=> mHCl = 0,2.36,5 = 7,3 (g)
=> \(m_{ddHCl}=\dfrac{7,3.100}{7,3}=100\left(g\right)\)
mdd sau pư = 0,1.24 + 100 - 0,1.2 = 102,2 (g)
\(C\%\left(MgCl_2\right)=\dfrac{0,1.95}{102,2}.100\%=9,2955\%\)
b)
CTHH: AaOb
PTHH: \(A_aO_b+2bHCl->aACl_{\dfrac{2b}{a}}+bH_2O\)
____________0,2------->\(\dfrac{0,1a}{b}\)
=> \(\dfrac{0,1a}{b}\left(M_A+35,5.\dfrac{2b}{a}\right)=13,5\)
=> \(M_A=\dfrac{64b}{a}=\dfrac{2b}{a}.32\)
Nếu \(\dfrac{2b}{a}=1\) => MA = 32 (L)
Nếu \(\dfrac{2b}{a}=2\) => MA = 64(Cu)

nFe = 11.2/56 = 0.2 (mol)
Fe + H2SO4 => FeSO4 + H2
0.2____0.2_______0.2___0.2
mH2SO4 = 0.2*98 = 19.6 (g)
mdd H2SO4 = 19.6*100/10 = 196 (g)
m dd sau phản ứng = 11.2 + 196 - 0.4 = 206.8 (g)
mFeSO4 = 0.2*152 = 30.4 (g)
C% FeSO4 = 30.4/206.8 * 100% = 14.7%
Vdd H2SO4 = mdd H2SO4 / D = 196 / 1.14 = 171.9 (ml)
CM FeSO4 = 0.2 / 0.1719 = 1.16 M

nFe = 11.2/56 = 0.2 (mol)
Fe + H2SO4 => FeSO4 + H2
0.2____0.2_______0.2___0.2
mH2SO4 = 0.2*98 = 19.6 (g)
mdd H2SO4 = 19.6*100/10 = 196 (g)
m dd sau phản ứng = 11.2 + 196 - 0.4 = 206.8 (g)
mFeSO4 = 0.2*152 = 30.4 (g)
C% FeSO4 = 30.4/206.8 * 100% = 14.7%
Vdd H2SO4 = mdd H2SO4 / D = 196 / 1.14 = 171.9 (ml)
CM FeSO4 = 0.2 / 0.1719 = 1.16 M
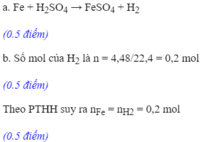

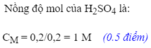
\(a,PTHH:Fe+H_2SO_4\to FeSO_4+H_2\\ b,n_{H_2}=\dfrac{16,8}{22,4}=0,74(mol)\\ \Rightarrow n_{Fe}=0,75(mol)\\ \Rightarrow m_{Fe}=0,75.56=42(g)\\ c,n_{H_2SO_4}=\dfrac{245.10\%}{100\%.98}=0,25(mol)\)
Vì \(\dfrac{n_{Fe}}{1}>\dfrac{n_{H_2SO_4}}{1}\) nên \(Fe\) dư
\(n_{Fe(dư)}=0,75-0,25=0,5(mol)\\ \Rightarrow m_{Fe(dư)}=0,5.56=28(g)\)