Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

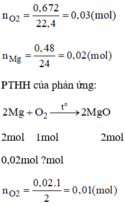
\(n_{Mg}=\dfrac{0.48}{24}=0.02\left(mol\right)\)
\(n_{O_2}=\dfrac{0.672}{22.4}=0.03\left(mol\right)\)
\(2Mg+O_2\underrightarrow{t^0}2MgO\)
\(0.02...0.01\)
\(3Fe+2O_2\underrightarrow{t^0}Fe_3O_4\)
\(0.03...\left(0.03-0.01\right)\)
\(m_{Fe}=0.03\cdot56=1.68\left(g\right)\)
\(m_{hh}=1.68+0.48=2.16\left(g\right)\)

Gọi x, y lần lượt là số mol của Fe và Mg.
Theo đề, ta có: \(56x+24y=13,2\) (*)
Ta có: \(n_{O_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
PTHH:
\(3Fe+2O_2\overset{t^o}{--->}Fe_3O_4\left(1\right)\)
\(2Mg+O_2\overset{t^o}{--->}2MgO\left(2\right)\)
Theo PT(1): \(n_{O_2}=\dfrac{2}{3}.n_{Fe}=\dfrac{2}{3}x\left(mol\right)\)
Theo PT(2): \(n_{O_2}=\dfrac{1}{2}.n_{Mg}=\dfrac{1}{2}y\left(mol\right)\)
\(\Rightarrow\dfrac{2}{3}x+\dfrac{1}{2}y=0,2\) (**)
Từ (*) và (**), ta có HPT:
\(\left\{{}\begin{matrix}56x+24y=13,2\\\dfrac{2}{3}x+\dfrac{1}{2}y=0,2\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}x=0,15\\y=0,2\end{matrix}\right.\)
\(\Rightarrow m_{Fe}=0,15.56=8,4\left(g\right)\)
\(m_{Mg}=0,2.24=4,8\left(g\right)\)

Gọi \(\left\{{}\begin{matrix}n_{Mg}=x\left(mol\right)\\x_{Ca}=y\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}24x+40y=17,6\\x=2y\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}x=0,4\\y=0,2\end{matrix}\right.\)
a)\(m_{Mg}=0,4\cdot24=9,6g\)
\(m_{Ca}=0,2\cdot40=8g\)
b)\(2Mg+O_2\underrightarrow{t^o}2MgO\)
\(2Ca+O_2\underrightarrow{t^o}2CaO\)
Từ hai pt: \(\Rightarrow\Sigma n_{O_2}=\dfrac{1}{2}n_{Mg}+\dfrac{1}{2}n_{Ca}=\dfrac{1}{2}\cdot0,4+\dfrac{1}{2}\cdot0,2=0,3mol\)
\(\Rightarrow m_{O_2}=0,3\cdot32=9,6g\)
\(V_{O_2}=0,3\cdot22,4=6,72l\)
\(\Rightarrow V_{kk}=5V_{O_2}=5\cdot6,72=33,6l\)

a)
Có \(\left\{{}\begin{matrix}24.n_{Mg}+40.n_{Ca}=17,6\\n_{Mg}=2.n_{Ca}\end{matrix}\right.\)
=> \(\left\{{}\begin{matrix}n_{Ca}=0,2\left(mol\right)\\n_{Mg}=0,4\left(mol\right)\end{matrix}\right.\)
=> \(\left\{{}\begin{matrix}m_{Ca}=0,2.40=8\left(g\right)\\m_{Mg}=0,4.24=9,6\left(g\right)\end{matrix}\right.\)
b)
PTHH: 2Ca + O2 --to--> 2CaO
0,2-->0,1
2Mg + O2 --to--> 2MgO
0,4--->0,2
=> \(V_{O_2}=\left(0,1+0,2\right).22,4=6,72\left(l\right)\)
\(V_{kk}=6,72.5=33,6\left(l\right)\)

PTHH: \(4Al+3O_2\underrightarrow{t^o}2Al_2O_3\) (1)
\(2Mg+O_2\underrightarrow{t^o}2MgO\) (2)
Ta có: \(\left\{{}\begin{matrix}n_{O_2\left(1\right)}=\dfrac{3}{4}n_{Al}=\dfrac{3}{4}\cdot\dfrac{13,5}{27}=0,375\left(mol\right)\\n_{O_2\left(1\right)}+n_{O_2\left(2\right)}=\dfrac{16,8}{22,4}=0,75\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow n_{O_2\left(2\right)}=0,375\left(mol\right)\) \(\Rightarrow n_{Mg}=0,75\left(mol\right)\)
\(\Rightarrow\%m_{Mg}=\dfrac{0,75\cdot24}{0,75\cdot24+13,5}\cdot100\%\approx57,14\%\)

a)
Theo ĐLBTKL: \(m_{Fe\left(bđ\right)}+m_{O_2}=m_X\)
=> \(m_{O_2}=26,4-20=6,4\left(g\right)\)
=> \(n_{O_2}=\dfrac{6,4}{32}=0,2\left(mol\right)\Rightarrow V=0,2.22,4=4,48\left(l\right)\)
b)
PTHH: 3Fe + 2O2 --to--> Fe3O4
0,2------->0,1
=> \(\%m_{Fe_3O_4}=\dfrac{0,1.232}{26,4}.100\%=87,88\%\)
c)
- Nếu dùng KClO3
PTHH: 2KClO3 --to--> 2KCl + 3O2
\(\dfrac{0,4}{3}\)<-----------------0,2
=> \(m_{KClO_3}=\dfrac{0,4}{3}.122,5=\dfrac{49}{3}\left(g\right)\)
- Nếu dùng KMnO4:
PTHH: 2KMnO4 --to--> K2MnO4 + MnO2 + O2
0,4<--------------------------------0,2
=> \(m_{KMnO_4}=0,4.158=63,2\left(g\right)\)

\(2Mg+O_2\xrightarrow{t^o}2MgO\\ 4Al+3O_2\xrightarrow{t^o}2Al_2O_3\\ \Rightarrow \begin{cases} 24.n_{Mg}+27.n_{Al}=5,1\\ 0,5.n_{Mg}+0,75.n_{Al}=n_{O_2}=\dfrac{2,8}{22,4}=0,125 \end{cases}\\ \Rightarrow \begin{cases} n_{Mg}=0,1(mol)\\ n_{Al_2O_3}=0,1(mol) \end{cases}\\ \Rightarrow \%m_{Mg}=\dfrac{0,1.24}{5,1}.100\%\approx 47,06\%\)
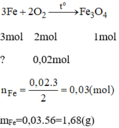
lối
ukm