Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a/ Ta có: \(n_{Mg}=\dfrac{4.8}{24}=0.2\left(mol\right)\)
PTHH:
\(2Mg+O_2\underrightarrow{t^o}2MgO\)
2 1
0.2 x
\(=>x=\dfrac{0.2\cdot1}{2}=0.1=n_{O_2}\)
\(=>V_{O_2\left(đktc\right)}=0.1\cdot22.4=2.24\left(l\right)\)
b/ \(2Mg+O_2\underrightarrow{t^o}2MgO\)
2 2
0.2 y
\(=>y=\left(0.2\cdot2\right):2=0.2=n_{MgO}\)
\(=>m_{MgO}=0.2\cdot\left(24+16\right)=8\left(g\right)\)

nO2 = 5,6/22,4 = 0,25 mol
Pt: 4Na + O2 -> 2Na2O (to)
1 mol<--0,25 mol
mNa cần dùng = 1 . 23 = 23 (g)
Chúc bạn học tốt ^_^
nO2 = \(\dfrac{5,6}{22,4}\)= 0,25 mol
ptpứ : 4Na + O2 → 2Na2O
1mol←0,25mol
khối lượng Na là :
mNa = 1. 23 = 23 g

\(a) 4P+ 5O_2 \xrightarrow{t^o} 2P_2O_5\\ b) n_{O_2} = \dfrac{1,12}{22,4} = 0,05(mol)\\ n_{P_2O_5} = \dfrac{2}{5}n_{O_2} = 0,02(mol)\\ m_{P_2O_5} = 0,02.142 = 2,84(gam) c) 2KClO_3 \xrightarrow{t^o} 2KCl + 3O_2\\ n_{KClO_3} = \dfrac{2}{3}n_{O_2} = \dfrac{0,1}{3}(mol)\\ m_{KClO_3} = \dfrac{0,1}{3}122,5 = 4,083(gam)\)

a) 4Al + 3O2 --to--> 2Al2O3
b) \(n_{O_2}=\dfrac{19,2}{32}=0,6\left(mol\right)\)
PTHH: 4Al + 3O2 --to--> 2Al2O3
0,8<-0,6---------->0,4
=> mAl = 0,8.27 = 21,6(g)
c) mAl2O3 = 0,4.102 = 40,8(g)

\(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\\a, PTHH:4Al+3O_2\rightarrow\left(t^o\right)2Al_2O_3\\ b,n_{O_2}=\dfrac{3}{4}.n_{Al}=\dfrac{3.0,2}{4}=0,15\left(mol\right)\\ \Rightarrow V_{O_2\left(đktc\right)}=0,15.22,4=3,36\left(l\right)\\ c,2KMnO_4\rightarrow\left(t^o\right)K_2MnO_4+MnO_2+O_2\\ n_{KMnO_4}=2.n_{O_2}=2.0,15=0,3\left(mol\right)\\ \Rightarrow m_{KMnO_4}=158.0,3=47,4\left(g\right)\)

\(n_{Zn}=\dfrac{m_{Zn}}{M_{Zn}}=\dfrac{19,5}{65}=0,3mol\)
\(Zn+\dfrac{1}{2}O_2\rightarrow\left(t^o\right)ZnO\)
1 1/2 1 (mol)
0,3 0,15 0,3 ( mol )
PƯ trên thuộc loại phản ứng hóa hợp
\(m_{ZnO}=n_{ZnO}.M_{ZnO}=0,3.81=24,3g\)
\(V_{O_2}=n_{O_2}.22,4=0,15.22,4=3,36l\)

a.4Al + 3O2 -> 2Al2O3
0.8 0.6 0.4
\(nO2=\dfrac{19.2}{32}=0.6mol\)
b.mAl = \(0.8\times27=21.6g\)
c.mAl2O3 = \(0.4\times102=40.8g\)

2Zn+O2-to>2ZnO
0,1---0,05----0,1
n Zn=0,1 mol
nO2=0,025 mol
=>VO2=0,05.22,4=1,12l
=>mZnO=0,1.81=8,1g
c)Zn dư
=>m ZnO=0,05.81=4,05g
2Zn+O2-to>2ZnO
0,1---0,05----0,1
n Zn=6,5/65=0,1 mol
n O2=0,8/32=0,025 mol
=>VO2=0,05.22,4=1,12l
=>mZnO=0,1.81=8,1g
c)Zn dư
=>m ZnO=0,05.81=4,05g

\(n_{Al}=\dfrac{5,4}{27}=0,2mol\)
\(4Al+3O_2\underrightarrow{t^o}2Al_2O_3\)
0,2 0,15 0,1
\(m_{Al_2O_3}=0,1\cdot102=10,2g\)
\(2KClO_3\underrightarrow{t^o}2KCl+3O_2\)
0,1 0,15
\(m_{KClO_3}=0,1\cdot122,5=12,25g\)
\(PTHH:4Al+3O_2\underrightarrow{t^o}2Al_2O_3\)
\(n_{Al}=5,4:27=0,2\left(mol\right)\)
\(\Rightarrow n_{Al_2O_3}=0,2.2:4=0,1\left(mol\right);n_{O_2}=0,2.3:4=0,15\left(mol\right)\)
\(m_{Al_2O_3}=0,1.102=10,2\left(g\right)\)
b)\(PTHH:2KClO_3\underrightarrow{t^o}2KCl+3O_2\)
\(n_{O_2}=0,15\left(mol\right)\)(câu a)
\(\Rightarrow n_{KClO_3}=0,15.2:3=0,1\left(mol\right)\)
\(m_{KClO_3}=0,1.123,5=12,35\left(g\right)\)
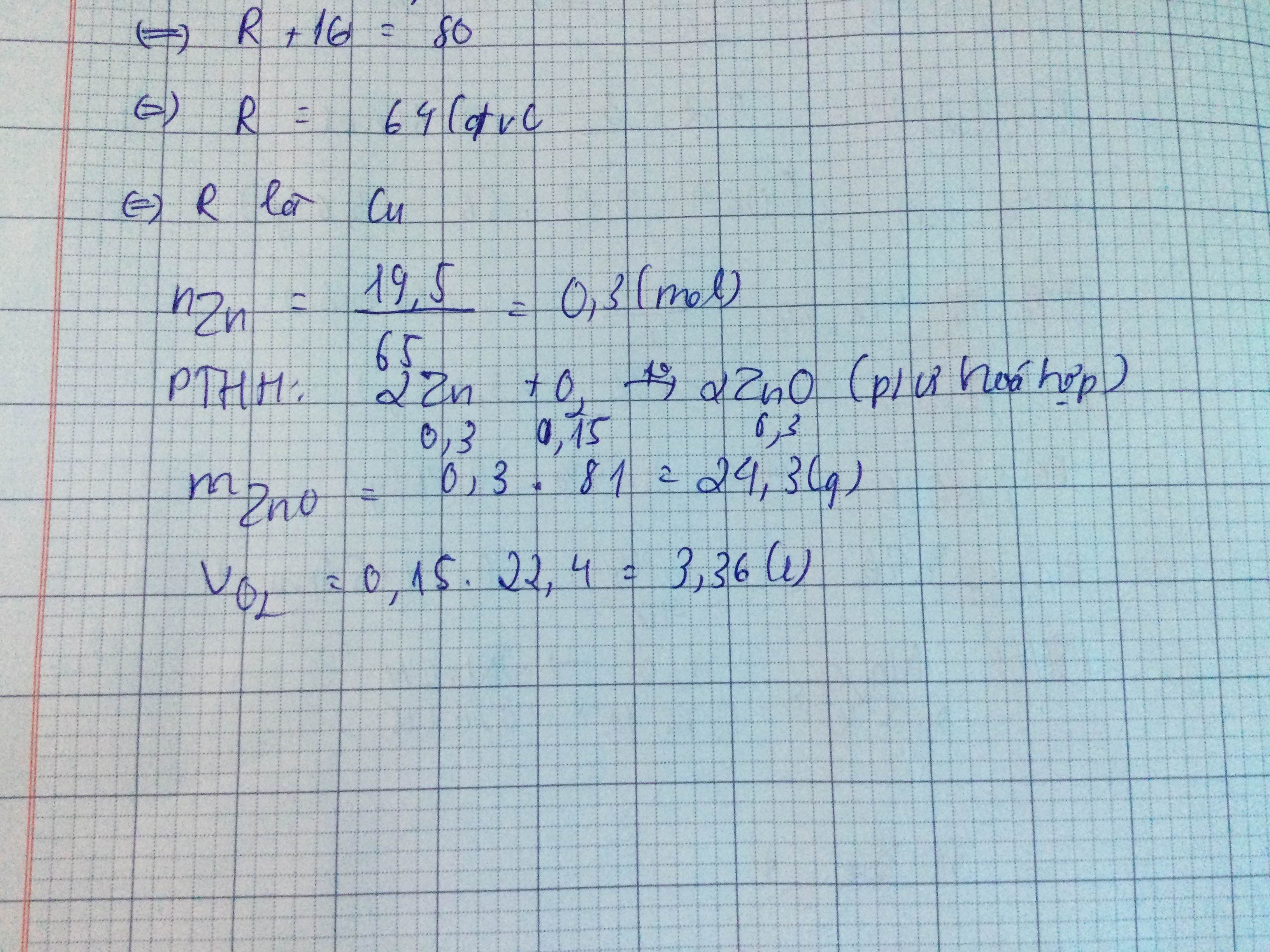
\(n_{Na_2O}=\dfrac{124}{62}=2\left(mol\right)\)
PTHH: 4Na + O2 --to--> 2Na2O
1<----------2
=> mO2 = 1.32 = 32 (g)
BTKLg
=>m O2=124-62=62g