Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

BT e: \(3n_{Al}+2n_{Mg}=4n_{O_2}+2n_{Cl_2}\)
\(\Rightarrow3\cdot0,1+2\cdot0,05=4\cdot0,05+2\cdot x\)
\(\Rightarrow x=0,1mol\)
\(m=m_{Al}+m_{Mg}+m_{O_2}+m_{Cl_2}\)
\(=0,1\cdot27+0,05\cdot24+0,05\cdot2\cdot16+0,1\cdot35,5\cdot2\)
\(=12,6g\)
Theo bảo toàn electron ta có: \(3\cdot n_{Al}+2\cdot n_{Mg}=2\cdot n_{Cl_2}+4\cdot N_{O_2}\)
\(\Rightarrow3\cdot0,1+2\cdot0,05=4\cdot0,05+2x\Rightarrow x=0,2\)
\(\Rightarrow m_Z=m_X+m_Y=0,1\cdot27+0,05\cdot24+0,05\cdot32+0,2\cdot71=19,7g\)

Ta có: \(n_{N_2O}+n_{NO_2}+n_{N_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\left(1\right)\)
BT e, có: 2nMg + 3nFe = 8nN2O + nNO2 + 10nN2
⇒ 8nN2O + nNO2 + 10nN2 = 1,9 (2)
Có: nHNO3 = 10nN2O + 2nNO2 + 12nN2 = 2,4.1 = 2,4 (mol) (3)
Từ (1), (2) và (3) \(\Rightarrow\left\{{}\begin{matrix}n_{N_2O}=0,1\left(mol\right)\\n_{NO_2}=0,1\left(mol\right)\\n_{N_2}=0,1\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\%V_{N_2}=\dfrac{0,1.22,4}{6,72.}100\%\approx33,33\%\)
\(n_{N_2O}=a\left(mol\right)\)
\(n_{NO_2}=b\left(mol\right)\)
\(n_{N_2}=c\left(mol\right)\)
\(\Rightarrow n_X=a+b+c=\dfrac{6.72}{22.4}=0.3\left(mol\right)\left(1\right)\)
Bảo toàn e :
\(8a+b+10c=0.65\cdot2+0.2\cdot3=1.9\left(2\right)\)
\(n_{H^+}=10a+2b+12c=2.4\left(mol\right)\left(3\right)\)
\(\left(1\right),\left(2\right),\left(3\right):a=b=c=0.1\left(mol\right)\)
\(\%V_{N_2}=\dfrac{0.1}{0.3}\cdot100\%=33.33\%\)

Ta có: nN2 = 0,22 (mol)
⇒ nNO3- = 10nN2 = 2,2 (mol)
⇒ m muối = mX + mNO3- = 31 + 2,2.62 = 167,4 (g)
nHNO3 = 12nN2 = 2,64 (mol)
\(\Rightarrow V_{HNO_3}=\dfrac{2,64}{2}=1,32\left(l\right)=1320\left(ml\right)\)

\(n_{HNO_3}=1.2\left(mol\right)\)
\(n_{NO}=a\left(mol\right)\)
\(n_{NO_2}=b\left(mol\right)\)
\(n_{N_2}=c\left(mol\right)\)
\(\Rightarrow a+b+c=\dfrac{5.6}{22.4}=0.25\left(1\right)\)
Bảo toàn e :
\(3\cdot0.2+1\cdot0.3=3a+b+10c\left(2\right)\)
\(n_{H^+}=4n_{NO}+2n_{NO_2}+12n_{N_2}\)
\(\Rightarrow4a+2b+12c=1.2\left(3\right)\)
\(\left(1\right),\left(2\right),\left(3\right):a=b=0.1,c=0.05\)
\(\%V_{NO_2}=\dfrac{0.1}{0.25}\cdot100\%=40\%\%0-\)

Ta có: \(n_{NO}+n_{NO_2}+n_{N_2}=\dfrac{22,4}{22,4}=1\left(mol\right)\left(1\right)\)
Mà: mX = 35,8 (g)
\(\Rightarrow30n_{NO}+46n_{NO_2}+28n_{N_2}=35,8\left(2\right)\)
Có: \(n_{Al}=\dfrac{32,4}{27}=1,2\left(mol\right)\)
\(n_{Cu}=\dfrac{22,4}{64}=0,35\left(mol\right)\)
BT e, có: 3nNO + nNO2 + 10nN2 = 3nAl + 2nCu = 4,3 (3)
Từ (1), (2) và (3) \(\Rightarrow\left\{{}\begin{matrix}n_{NO}=0,3\left(mol\right)\\n_{NO_2}=0,4\left(mol\right)\\n_{N_2}=0,3\left(mol\right)\end{matrix}\right.\)
⇒ nHNO3 = 4nNO + 2nNO2 + 12nN2 = 5,6 (mol)

Ta có: \(n_{N_2O}+n_{NO_2}+n_{N_2}=\dfrac{11,2}{22,4}=0,5\left(mol\right)\left(1\right)\)
\(n_{HNO_3}=1,85.2=3,7\left(mol\right)\)
⇒ 10nN2O + 2nNO2 + 12nN2 = 3,7 (2)
\(n_{Mg}=\dfrac{16,8}{24}=0,7\left(mol\right)\)
\(n_{Fe}=\dfrac{28}{56}=0,5\left(mol\right)\)
BT e, có: 8nN2O + nNO2 + 10nN2 = 2nMg + 3nFe = 2,9 (3)
Từ (1), (2) và (3) \(\Rightarrow\left\{{}\begin{matrix}n_{N_2O}=0,15\left(mol\right)\\n_{NO_2}=0,2\left(mol\right)\\n_{N_2}=0,15\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\%V_{N_2}=\dfrac{0,15}{0,5}.100\%=30\%\)
m muối = mMg + mFe + 62.(8nN2O + nNO2 + 10nN2) = 224,6 (g)

a) Sửa đề: dd H2SO4 9,8%
Ta có: \(n_{H_2}=\dfrac{7,84}{22,4}=0,35\left(mol\right)\) \(\Rightarrow m_{H_2}=0,35\cdot2=0,7\left(g\right)\)
Bảo toàn nguyên tố: \(n_{H_2SO_4}=n_{H_2}=0,35\left(mol\right)\) \(\Rightarrow m_{ddH_2SO_4}=\dfrac{0,35\cdot98}{9,8\%}=350\left(g\right)\)
\(\Rightarrow m_{dd}=m_{KL}+m_{H_2SO_4}-m_{H_2}=361,6\left(g\right)\)
b) Tương tự câu a

Đáp án A.
![]()
![]()
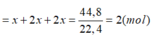
![]()

Bảo toàn e ta có: 3x = 1,2 + 6,4 + 8 = 15,6 => x =5,2 .
mAl = 5,2.27=140,4(gam).
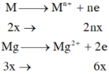
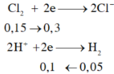
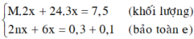
a, Gọi: \(\left\{{}\begin{matrix}n_{Mg}=x\left(mol\right)\\n_{Al}=2y\left(mol\right)\\n_{Zn}=y\left(mol\right)\end{matrix}\right.\)
⇒ 24x + 27.2y + 65y = 19,1 (1)
BT e, có: 2nMg + 3nAl + 2nZn = 10nN2 + 8nN2O
⇒ 2x + 3.2y + 2y = 10.0,1 + 8.0,05 (2)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}x=0,3\left(mol\right)\\y=0,1\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\%m_{Mg}=\dfrac{0,3.24}{19,1}.100\%\approx37,7\%\)
b, Ta có: nHNO3 = 12nN2 + 10nN2O = 1,7 (mol)
\(\Rightarrow C_{M_{HNO_3}}=\dfrac{1,7}{2}=0,85\left(M\right)=x\)