
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


(1) C + O 2 → t ° C O 2
(2) C O 2 + Ca OH 2 → CaC O 3 + H 2 O
(3) CaC O 3 → CaO + C O 2
(4) CaO + H 2 O → Ca OH 2
(5) Ca OH 2 + 2C O 2 → Ca HCO 3 2

(1) Fe 2 O 3 + 3 H 2 → 2Fe + 3 H 2 O
(2) 2Fe + 3 Cl 2 → 2Fe Cl 3
(3) Fe Cl 3 + 3NaOH → 3NaCl + Fe OH 3
(4) 2Fe OH 3 → Fe 2 O 3 + 3 H 2 O
(5) Fe + HCl → Fe Cl 2 + H 2
(6) Fe Cl 2 + 2NaOH → Fe OH 2 + 2NaCl

\(Na_2O+H_2O\rightarrow2NaOH\\ 2NaOH+CO_2\rightarrow Na_2CO_3+H_2O\\ Na_2CO_3+2HCl\rightarrow2NaCl+CO_2+H_2O\\ CO_2+H_2O\rightarrow H_2CO_3\)

Sửa đề: C2H3OH thành C2H5OH
\(C_2H_4+H_2O\overrightarrow{axit}C_2H_5OH\\ C_2H_5OH+O_2\underrightarrow{\text{men giấm}}CH_3COOH\\ C_2H_5OH+CH_3COOH\xrightarrow[H_2SO_4đặc]{t^o}CH_3COOC_2H_5+H_2O\)

a.
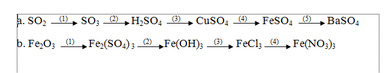
\(1.2SO_2+O_2->2SO_3\)
\(2.H_2O+SO_3->H_2SO_4\)
\(3.CuO+H_2SO_4->CuSO_4+H_2O\)
\(4.Fe+CuSO_4->FeSO_4+Cu\)
\(5.FeSO_4+BaCl_2->BaSO_4+FeCl_2\)
a.
\(\left(1\right)2SO_2+O_2\xrightarrow[V_2O_5]{t^o}2SO_3\)
\(\left(2\right)SO_3+H_2O\xrightarrow[]{}H_2SO_4\)
\(\left(3\right)H_2SO_4+CuO\xrightarrow[]{}CuSO_4+H_2O\)
\(\left(4\right)Fe+CuSO_4\xrightarrow[]{}Cu\downarrow+FeSO_4\)
\(\left(5\right)FeSO_4+BaCl_2\xrightarrow[]{}BaSO_4\downarrow+FeCl_2\)
b.
\(\left(1\right)Fe_2O_3+3H_2SO_4\xrightarrow[]{}Fe_2\left(SO_4\right)_3+3H_2O\)
\(\left(2\right)Fe_2\left(SO_4\right)_3+6NaOH\xrightarrow[]{}2Fe\left(OH\right)_3+3Na_2SO_4\)
\(\left(3\right)Fe\left(OH\right)_3+3HCl\xrightarrow[]{}FeCl_3+3H_2O\)
\(\left(4\right)FeCl_3+3HNO_3\xrightarrow[]{}Fe\left(NO_3\right)_3+3HCl\)

\(VD:Fe\rightarrow FeCl_3\\ PTHH:2Fe+3Cl_2\rightarrow\left(t^o\right)2FeCl_3\\ VD2:NaCl\rightarrow Na\\ PTHH:2NaCl\rightarrow\left(đpnc\right)2Na+Cl_2\)

\(C_2H_4+H_2O\underrightarrow{t^o,xt}C_2H_5OH\)
\(C_2H_5OH+O_2\underrightarrow{^{mengiam}}CH_3COOH+H_2O\)
\(CH_3COOH+C_2H_5OH⇌CH_3COOC_2H_5+H_2O\) (xt: H2SO4 đặc, to)
\(CH_3COOC_2H_5+NaOH\underrightarrow{t^o}CH_3COONa+C_2H_5OH\)
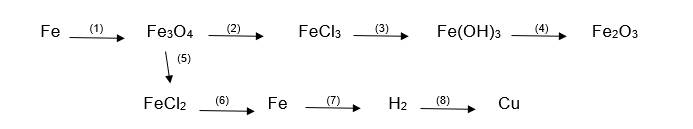
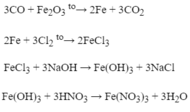

(1) \(3Fe+2O_2\xrightarrow[]{t^\circ}Fe_3O_4\)
(2) \(Fe_3O_4+8HCl\xrightarrow[]{}FeCl_2+2FeCl_3+4H_2O\)
(3) \(FeCl_3+3LiOH\xrightarrow[]{}Fe\left(OH\right)_3\downarrow+3LiCl\)
(4) \(2Fe\left(OH\right)_3\xrightarrow[]{t^\circ}Fe_2O_3+3H_2O\)
(5) \(Fe_3O_4+8HCl\xrightarrow[]{}FeCl_2+2FeCl_3+4H_2O\)
(6) \(FeCl_2\xrightarrow[]{đpdd}Fe+Cl_2\uparrow\)
(7) \(Fe+2HCl\xrightarrow[]{}FeCl_2+H_2\uparrow\)
(8) \(H_2+CuO\xrightarrow[]{t^\circ}Cu+H_2O\uparrow\)
(1) 3��+2�2→�∘��3�43Fe+2O2t∘Fe3O4
(2) ��3�4+8���→����2+2����3+4�2�Fe3O4+8HClFeCl2+2FeCl3+4H2O
(3) ����3+3����→��(��)3↓+3����FeCl3+3LiOHFe(OH)3↓+3LiCl
(4) 2��(��)3→�∘��2�3+3�2�2Fe(OH)3t∘Fe2O3+3H2O
(5) ��3�4+8���→����2+2����3+4�2�Fe3O4+8HClFeCl2+2FeCl3+4H2O
(6) ����2→đ�����+��2↑FeCl2đpddFe+Cl2↑
(7) ��+2���→����2+�2↑Fe+2HClFeCl2+H2↑
(8) �2+���→�∘��+�2�↑H2+CuOt∘Cu+H2O↑