Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a)
$V_{O_2} = 0,2.22,4 = 4,48(lít)$
b)
$n_{SO_2} = \dfrac{76,8}{64} = 1,2(mol)$
$V_{SO_2} = 1,2.22,4 = 26,88(lít)$
c)
$n_{N_2} = \dfrac{7,5.10^{23}}{6.10^{23}} = 1,25(mol)$
$V_{N_2} = 1,25.22,4 = 28(lít)$
d)
$V_X = (0,2 + 0,25).22,4 = 10,08(lít)$
a) V(O2,đktc)=0,2.22,4=4,48(l)
b) V(SO2,đktc)= (76,8/64).22,4=26,88(l)
c) V(N2,đktc)= [(7,5.1023)/(6.1023)].22,4= 28(l)
d) VhhX(đktc)= V(O2,đktc) + V(N2,đktc)= 0,2.22,4+0,25.22,4=10,08(l)

\(a.V_{N_2}=n.22,4=0,25.22,4=5,6\left(l\right)\)
\(b.n_{NH_3}=\dfrac{0,9.10^{23}}{6.10^{23}}=0,15\left(mol\right)\\ V_{NH_3}=n.22,4=0,15.22,4=3,36\left(l\right)\)
\(c.n_{SO_2}=\dfrac{3,2}{64}=0,05\left(mol\right)\\ V_{SO_2}=n.22,4=0,05.22,4=1,12\left(l\right)\)

\(1,\\ a,m_{hh}=3.44+2.28=188(g)\\ b,m_{hh}=\dfrac{2,24}{22,4}.64+\dfrac{1,12}{22,4}.32=8(g)\\ 2,\\ a,V_{hh}=(\dfrac{4,4}{44}+\dfrac{0,4}{2}).22,4=6,72(l)\\ b,V_{hh}=(\dfrac{6.10^{23}}{6.10^{23}}+\dfrac{3.10^{23}}{6.10^{23}}).22,4=33,6(l)\)

a) \(n_{NaOH}=\dfrac{8}{40}=0,2\left(mol\right)\)
b) \(n_{N_2}=\dfrac{1,8.10^{23}}{6.10^{23}}=0,3\left(mol\right)\)
=> \(m_{N_2}=0,3.28=8,4\left(g\right)\)
c) \(n_{CO_2}=\dfrac{8,8}{44}=0,2\left(mol\right)=>V_{CO_2}=0,2.22,4=4,48\left(l\right)\)
d) \(n_{H_2}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\)
=> Số phân tử H2 = 0,15.6.1023 = 0,9.1023
e) \(n_{O_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
f) \(n_{Cl_2}=\dfrac{3,6.10^{23}}{6.10^{23}}=0,6\left(mol\right)\)
=> VCl2 = 0,6.22,4 = 13,44(l)
g) \(n_{O_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
=> mO2 = 0,3.32 = 9,6(g)
h) \(n_{K_2O}=\dfrac{18,8}{94}=0,2\left(mol\right)\)
=> Số phân tử K2O = 0,2.6.1023 = 1,2.1023
i) \(n_{CaO}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
=> Số phân tử CaO = 0,2.6.1023 = 1,2.1023
nHCl = 0,2.1,5 = 0,3 (mol)
=> mHCl = 0,3.36,5 = 10,95(g)

a, VO\(_2\) = 0,15 . 22,4 = 3,36 lít
b, V\(CO_2\) = \((\dfrac{48}{44}).22,4\approx24,43\) ( lít )
c, \(V_{SO_2}=\left(\dfrac{16}{64}\right).22,4=5,6\) ( lít )
\(V_{H_2}=\left(\dfrac{18.10^{23}}{6.10^{23}}\right).22,4=67,2\) ( lít )
=> \(V_{hh}=5,6+67,2=72,8\) ( lít )

\(a_1,m_{CaCO_3}=0,25.100=25(g)\\ a_2,m_{SO_2}=\dfrac{3,36}{22,4}.64=9,6(g)\\ a_3,m_{H_2SO_4}=\dfrac{9.10^{23}}{6.10^{23}}.98=147(g)\)
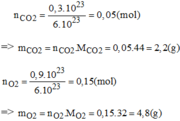
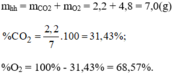
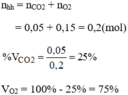
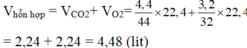

\(a.n_{CO_2}=\dfrac{m_{CO_2}}{M_{CO_2}}=\dfrac{4,4}{44}=0,1\left(mol\right)\\ \Rightarrow V_{CO_2}=n_{CO_2}.22,4=0,1.22,4=2,24\left(l\right)\\ n_{O_2}=\dfrac{m_{O_2}}{M_{O_2}}=\dfrac{3,2}{32}=0,1\left(mol\right)\)
\(\Rightarrow V_{O_2}=n_{O_2}.22,4=0,1.22,4=2,24\left(l\right)\)
\(n_{H_2}\) = 2mol
=>\(V_{H_2}\) = 2. 22,4 = 44,8l
\(n_{O_2}\) = 0,0875 mol
=>\(V_{O_2}\) = 0,0875 . 22,4 = 1,96l
\(n_{CO_2}\) = 0,5 mol
=>\(V_{CO_2}\) = 0,5 .22,4 = \(11,2\left(l\right)\)
\(n_{O_2}\) = \(0,2\left(mol\right)\)
=>\(V_{O_2}\) = 0,2 . 22,4 = \(4,48\left(l\right)\)
b) Ta có:
\(n_{NH_3}=\dfrac{6.10^{23}}{6.10^{23}}=1\left(mol\right)\\ n_{O_2}=\dfrac{3.10^{23}}{6.10^{23}}=0,5\left(mol\right)\\ \Sigma n_{hh}=1+0,5=1,5\left(mol\right)\\ V_{hh}=1,5.22,4=33,6\left(l\right)\)