Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(CaCO_3+2CH_3COOH\rightarrow\left(CH_3COO\right)_2Ca+CO_2+H_2O\\ a,n_{CH_3COOH}=0,2.1=0,2\left(mol\right)\Rightarrow n_{CO_2}=\dfrac{0,2}{2}=0,1\left(mol\right)\Rightarrow V_{CO_2\left(đktc\right)}=0,1.22,4=2,24\left(l\right)\\ b,CH_3COOH+C_2H_5OH⇌\left(H^+,t^o\right)CH_3COOC_2H_5+H_2O\\ n_{CH_3COOH}=\dfrac{50}{200}.0,2=0,05\left(mol\right)\\ n_{C_2H_5OH}=\dfrac{23}{46}=0,5\left(mol\right)\\ Vì:\dfrac{0,5}{1}>\dfrac{0,05}{1}\Rightarrow Rượu.dư\\ \Rightarrow n_{este\left(LT\right)}=n_{axit}=0,05\left(mol\right)\\ \Rightarrow n_{este\left(TT\right)}=80\%.0,05=0,04\left(mol\right)\\ m_{CH_3COOC_2H_5}=88.0,04=3,52\left(g\right)\)

F e + 2 H C l → F e C L 2 + H 2
Vì H = 85% nên:
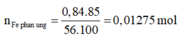
n H 2 = n F e p ư = 0,01275 mol
V H 2 = 0,01275.22,4= 0,2856 lit
⇒ Chọn C.

2H2+O2-to>2H2O
0,4----0,2------0,4 mol
n O2=\(\dfrac{4,48}{22,4}\)=0,2 mol
=>VH2=0,4.22,4=8,96l
=>m H2O=0,4.18=7,2g
c) ta có :mdd HCl =m H2O+m HCl=50g
=>m HCl=50-7,2=42,8g
=>C%HCl=\(\dfrac{42,8}{50}.100\)=85,6%
a) \(n_{O_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
PTHH: 2H2 + O2 --to--> 2H2O
0,4<--0,2-------->0,4
=> \(V_{H_2}=0,4.22,4=8,96\left(l\right)\)
b) \(m_{H_2O}=0,4.18=7,2\left(g\right)\)
c) mHCl = 50 - 7,2 = 42,8 (g)
=> \(C\%_{ddHCl}=\dfrac{42,8}{50}.100\%=85,6\%\)
không bt sai đâu không nhưng nồng độ cao nhất của HCl ở khoảng 40% nhé :)

Ta có: \(n_{CaCO_3}=\dfrac{20}{100}=0,2\left(mol\right)\)
PT: \(CaCO_3+2HCl\rightarrow CaCl_2+CO_2+H_2O\)
______0,2_______0,4_____0,2____0,2 (mol)
a, \(m_{ddHCl}=\dfrac{0,4.36,5}{14,6\%}=100\left(g\right)\)
b, \(V_{CO_2}=0,2.22,4=4,48\left(l\right)\)
c, m dd sau pư = 20 + 100 - 0,2.44 = 111,2 (g)
\(\Rightarrow C\%_{CaCl_2}=\dfrac{0,2.111}{111,2}.100\%\approx19,96\%\)

1)
\(n_{Al}=\dfrac{2,7}{27}=0,1\left(mol\right)\)
PTHH: 2Al + 3H2SO4 --> Al2(SO4)3 + 3H2
____0,1----->0,15
=> mH2SO4 = 0,15.98 = 14,7(g)
=> \(C\%=\dfrac{14,7}{250}.100\%=5,88\%\)
2)
\(n_{Na_2CO_3}=\dfrac{21,2}{106}=0,2\left(mol\right)\)
PTHH: Na2CO3 + 2HCl --> 2NaCl + CO2 + H2O
_______0,2------------------------------>0,2
=> VCO2 = 0,2.22,4 = 4,48(l)
3)
\(n_A=\dfrac{18,4}{M_A}\left(mol\right)\)
PTHH: 2A + Cl2 --to--> 2ACl
____\(\dfrac{18,4}{M_A}\)---------->\(\dfrac{18,4}{M_A}\)
=> \(\dfrac{18,4}{M_A}\left(M_A+35,5\right)=46,8=>M_A=23\left(Na\right)\)
4)
nHCl = 0,2.3 = 0,6(mol)
PTHH: M + 2HCl --> MCl2 + H2
____0,3<-----0,6
=> \(M_M=\dfrac{7,2}{0,3}=24\left(Mg\right)\)

\(a.n_{CO_2}=\dfrac{0,672}{22,4}=0,03mol\\ CO_2+Ca\left(OH\right)_2\rightarrow CaCO_3+H_2O\)
\(n_{CO_2}=n_{Ca\left(OH\right)_2}=n_{CaCO_3}=0,03mol\\ m_{CaCO_3}=0,03.100=3g\\ b.V_{ddCa\left(OH\right)_2}=\dfrac{0,03}{1,5}=0,02l\)

PTHH: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\uparrow\)
Ta có: \(n_{HCl}=0,18\cdot1=0,18\left(mol\right)\)
\(\Rightarrow n_{H_2\left(LT\right)}=0,09\left(mol\right)\)
\(\Rightarrow H\%=\dfrac{\dfrac{1,512}{22,4}}{0,09}\cdot100\%=75\%\)
PT: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
Ta có: \(n_{HCl}=0,18.1=0,18\left(mol\right)\)
Theo PT: \(n_{H_2\left(LT\right)}=\dfrac{1}{2}n_{HCl}=0,09\left(mol\right)\)
\(\Rightarrow V_{H_2\left(LT\right)}=0,09.22,4=2,016\left(l\right)\)
Mà: VH2 (TT) = 1,512 (l)
\(\Rightarrow H\%=\dfrac{1,512}{2,016}.100\%=75\%\)
Bạn tham khảo nhé!



CaCO3+2HCl----->CaCl2+H2O+CO2
Cứ 1 mol CaCO3------->1 mol CO2
100g------->22,4 l
50g--------->11,2L
mà H%=85%------>VCO2=11,2.85/100=9,52(l)