Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(Fe+2HCl\rightarrow FeCl_2+H_2\\ n_{Fe}=n_{H_2}=\dfrac{9,9161}{24,79}\approx0,4\left(mol\right)\\ b,m_{Fe}\approx0,4.56\approx22,4\left(g\right)\\ c,n_{HCl}\approx0,4.2\approx0,8\left(mol\right)\\ C_{MddHCl}\approx\dfrac{0,8}{0,25}\approx3,2\left(M\right)\)

\(n_{H_2}=\dfrac{3,36}{22,4}=0,15(mol)\\ PTHH:Fe+H_2SO_4\to FeSO_4+H_2\\ \Rightarrow n_{H_2SO_4}=n_{H_2}=0,15(mol)\\ \Rightarrow C_{M_{H_2SO_4}}=\dfrac{0,15}{0,05}=3M\\ PTHH:2H_2+O_2\xrightarrow{t^o}2H_2O\\ \Rightarrow n_{O_2}=\dfrac{1}{2}n_{H_2}=0,075(mol)\\ \Rightarrow V_{O_2}=0,075.22,4=1,68(l)\)

Fe+2HCl->FeCl2+H2
0,3----0,6--0,3------0,3
n H2=0,3 mol
=>m Fe=0,3.56=16.8g
=>CM=0,6\0,1=6M
CuO+H2-tO>Cu+H2O
0.3---------------0,3
=>m Cu=0,3.64=19,2g

a) Fe+H2SO4------>FeSO4+H2
b)nH2=5,6/22,4=0,25mol
Fe+H2SO4------->FeSO4+H2
0,25 <---- 0,25
Theo pt:
nFe=0,25mol
=>mFe=0,25×56=14g
c)
Fe+H2SO4------->FeSO4+H2
0,25 <---- 0,25
Theo pt:
nH2SO4=0,25 mol
CM=n/V=0,25/0,05=5(M)
\(n_{H_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\)
\(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
0,25 .... 0,25 ...... 0,25 ..... 0,25 (mol)
a. \(m_{Fe}pư=0,25.56=14\left(g\right)\)
b. \(C_M\left(H_2SO_4\right)=C_M\left(FeSO_4\right)=\dfrac{0,25}{0,05}=5\left(M\right)\)

a, \(Fe+2HCl\rightarrow FeCl_2+H_2\)
b, \(n_{H_2}=\dfrac{9,916}{24,79}=0,4\left(mol\right)\)
\(n_{Fe}=n_{H_2}=0,4\left(mol\right)\Rightarrow m_{Fe}=0,4.56=22,4\left(g\right)\)
c, \(n_{HCl}=2n_{H_2}=0,8\left(mol\right)\Rightarrow C_{M_{HCl}}=\dfrac{0,8}{0,2}=4\left(M\right)\)

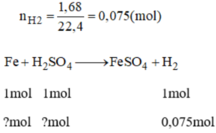
Theo phương trình phản ứng ta có:
n F e = n H 2 = 0,075 mol
n H 2 S O 4 = 0,075 mol (mà H 2 S O 4 đề cho là 0,2 mol nên H 2 S O 4 dư)
![]()

Sửa lại câu c .
\(n_{H_2SO_4}=\dfrac{49.40}{100}:98=0,2\left(mol\right)\)
\(PTHH:\)
\(Fe+H_2SO_4\rightarrow FeSO_4+H_2\uparrow\)
trc p/u : 0,3 0,2
p/u : 0,2 0,2 0,2 0,2
sau : 0,1 0 0,2 0,2
-> Fe dư
\(m_{ddFeSO_4}=0,3.56+49-0,4=65,4\left(g\right)\) ( ĐLBTKL )
\(m_{FeSO_4}=0,2.152=30,4\left(g\right)\)
\(C\%=\dfrac{30,4}{65,4}.100\%\approx46,48\%\)
PTHH :
\(Fe+H_2SO_4\rightarrow FeSO_4+H_2\uparrow\)
0,3 0,3 0,3 0,3
\(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
\(a,m_{Fe}=0,3.56=16,8\left(g\right)\)
\(b,C_M=\dfrac{n}{V}=\dfrac{0,3}{0,2}=1,5M\)
\(c,n_{H_2SO_4}=\dfrac{\dfrac{49.40}{100}}{98}=0,2\left(mol\right)\)
\(\rightarrow n_{FeSO_4}=n_{H_2SO_4}=0,2\left(mol\right)\)
\(m_{FeSO_4}=0,2.152=30,4\left(g\right)\)
\(m_{ddFeSO_4}=49+\left(0,2.56\right)-0,2.2=59,8\left(g\right)\)( định luật bảo toàn khối lượng )
\(C\%=\dfrac{30,4}{59,8}.100\%\approx50,84\%\)

\(n_{H_2SO_4}=\dfrac{200.20\%}{98}=\dfrac{20}{49}\left(mol\right)\\a, PTHH:Fe+H_2SO_4\rightarrow FeSO_4+H_2\\ n_{Fe}=n_{H_2}=n_{H_2SO_4}=\dfrac{20}{49}\left(mol\right)\\ b,m_{Fe}=\dfrac{20}{49}.56=\dfrac{160}{7}\left(g\right)\\ c,V_{H_2\left(đktc\right)}=\dfrac{20}{49}.22,4=\dfrac{64}{7}\left(l\right)\)
a, PT: \(Fe+2H_2SO_4\rightarrow FeSO_4+H_2\)
b, \(m_{H_2SO_4}=200.20\%=40\left(g\right)\Rightarrow n_{H_2SO_4}=\dfrac{40}{98}=\dfrac{20}{49}\left(mol\right)\)
Theo PT: \(n_{Fe}=n_{H_2SO_4}=\dfrac{20}{49}\left(mol\right)\Rightarrow m_{Fe}=\dfrac{20}{49}.56=\dfrac{160}{7}\left(g\right)\)
c, \(n_{H_2}=n_{H_2SO_4}=\dfrac{20}{49}\left(mol\right)\Rightarrow V_{H_2}=\dfrac{20}{49}.22,4=\dfrac{64}{7}\left(l\right)\)
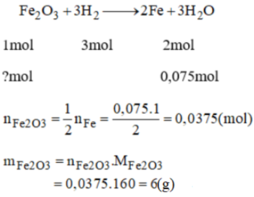
a) \(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
b) \(n_{H_2}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\)
Theo PTHH: \(n_{Fe}=n_{H_2}=0,15\left(mol\right)\)
\(\Rightarrow m_{Fe}=0,15\cdot56=8,4\left(g\right)\)
c) Theo PTHH: \(n_{H_2SO_4}=n_{H_2}=0,15\left(mol\right)\)
\(V_{H_2SO_4}=50ml=0,05\left(l\right)\)
\(\Rightarrow C_{M,H_2SO_4}=\dfrac{n_{H_2SO_4}}{V_{H_2SO_4}}=\dfrac{0,15}{0,05}=3M\)