Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(2Al+6HCl->2AlCl_3+3H_2\\ Fe+2HCl->FeCl_2+H_2\\ n_{Al}=a;n_{Fe}=b\\ 27a+56b=8,3\\ 1,5a+b=\dfrac{5,6}{22,4}=0,25\\ a=b=0,1\\ m_{Al}=27\cdot0,1=2,7g\\ m_{Fe}=8,3-2,7=5,6g\\ a=\dfrac{3a+2b}{500}\cdot36,5=3,65\%\\ m_{ddsau}=508,3-0,25\cdot2=507,8g\\ C\%_{AlCl_3}=\dfrac{133,5a}{507,8}=2,63\%\\ C\%_{FeCl_2}=\dfrac{127b}{507,8}=2,50\%\)

Gọi số mol Al, Fe là a, b (mol)
=> 27a + 56b = 11,1 (1)
\(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
a----------------------->1,5a
Fe + 2HCl --> FeCl2 + H2
b------------------------>b
=> 1,5a + b = 0,3 (2)
(1)(2) => a = 0,1; b = 0,15
=> \(\left\{{}\begin{matrix}\%m_{Al}=\dfrac{0,1.27}{11,1}.100\%=24,32\%\\\%m_{Fe}=\dfrac{0,15.56}{11,1}.100\%=75,68\end{matrix}\right.\)

\(n_{H_2}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\)
PTHH: Fe + 2HCl --> FeCl2 + H2
0,15<--------------------0,15
=> \(\%Fe=\dfrac{0,15.56}{14,8}.100\%=56,757\%\)
=> \(\%Cu=100\%-56,757\%=43,243\%\)
nH2 = 3,36/22,4 = 0,15 (mol)
PTHH: Fe + 2HCl -> FeCl2 + H2
nFe = 0,15 (mol)
mFe = 0,15 . 56 = 8,4 (g)
%mFe = 8,4/14,8 = 56,75%
%mCu = 100% - 56,75% = 43,25%

a)
Gọi : \(\left\{{}\begin{matrix}n_{Al}=a\left(mol\right)\\n_{Fe}=b\left(mol\right)\end{matrix}\right.\)⇒ 27a + 56b = 1,66(1)
\(2Al + 6HCl \to 2AlCl_3 + 3H_2\\ Fe +2 HCl \to FeCl_2 + H_2\)
Theo PTHH :
\(n_{H_2} = 1,5a + b = \dfrac{1,12}{22,4} = 0,05(2)\)
Từ (1)(2) suy ra a = 0,02 ; b = 0,02
Vậy :
\(\%m_{Al} = \dfrac{0,02.27}{1,66}.100\% = 32,53\%\\ \%m_{Fe} = 100\% - 32,53\% = 67,47\%\)
a)
\(n_{HCl} = 2n_{H_2} = 0,05.2 = 0,1(mol)\\ \Rightarrow C\%_{HCl} = \dfrac{0,1.36,5}{100}.100\% = 3,65\%\)

Fe+2HCl->FeCl2+H2
x---2x-----------x
Mg+2HCl->MgCl2+H2
y------2y-----------y
Ta có :
\(\left\{{}\begin{matrix}56x+24y=24\\x+y=\dfrac{13,44}{22,4}\end{matrix}\right.\)
=>x=0,3 mol, y=0,3 mol
=>%m Fe=\(\dfrac{0,3.56}{24}.100\)=70%
=>%m Mg=100-70=30%
=>VHCl=\(\dfrac{0,3.2+0,3.2}{2}\)=0,6l=600ml
b)
XCl2+2AgNO3->2AgCl+X(NO3)2
0,6--------------------1,2mol
=>m AgCl=1,2.143,5=172,2g

\(Fe+2HCl\rightarrow FeCl_2+H_2\\ n_{Fe}=n_{H_2}=\dfrac{0,448}{22,4}=0,02\left(mol\right)\\ a,\%m_{Fe}=\dfrac{0,02.56}{4,36}.100\approx25,688\%\\ \Rightarrow\%m_{Ag}\approx74,312\%\\ b,Ta.thấy:2,18=\dfrac{1}{2}.4,36\\ \Rightarrow m_{hh\left(câuB\right)}=\dfrac{1}{2}.m_{hh\left(câuA\right)}\\ n_{Fe}=\dfrac{0,02}{2}=0,01\left(mol\right)\\ n_{Ag}=\dfrac{2,18-0,01.56}{108}=0,015\left(mol\right)\\ 2Fe+3Cl_2\rightarrow\left(t^o\right)2FeCl_3\\ 2Ag+Cl_2\rightarrow\left(t^o\right)2AgCl\\ n_{Cl_2}=\dfrac{3}{2}.n_{Fe}+\dfrac{1}{2}.n_{Ag}=\dfrac{3}{2}.0,01+\dfrac{1}{2}.0,015=0,0225\left(mol\right)\\ \Rightarrow V_{Cl_2\left(đktc\right)}=0,0225.22,4=0,504\left(l\right)\)

Câu 1:
Gọi số mol Al là x; Zn là y
\(\rightarrow27x+65y=18,4\)
\(Al+3HCl\rightarrow AlCl_3+\frac{3}{2}H_2\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
\(\rightarrow n_{H2}=1,5n_{Al}+n_{Zn}=1,5x+y=\frac{1}{2}=0,5\left(mol\right)\)
Giải được: \(x=y=0,2\)
\(\Rightarrow m_{Al}=27x=5,4\left(g\right)\Rightarrow\%m_{Al}=\frac{5,4}{18,4}=29,3\%\Rightarrow\%m_{Zn}=70,7\%\)Câu 2:
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(FeO+2HCl\rightarrow FeCl_2+H_2\)
Ta có: \(n_{H2}=n_{Fe}=\frac{2,24}{22,4}=0,1\left(mol\right)\)
Muối thu được là FeCl2
\(\rightarrow n_{FeCl2}=\frac{38,1}{56+35,5.2}=0,3\left(mol\right)\)
Ta có: \(n_{FeCl2}=n_{Fe}+n_{FeO}\rightarrow n_{FeO}=0,2\left(mol\right)\)
\(\Rightarrow m_{Fe}=0,1.56=5,6\left(g\right);m_{FeO}=0,2.\left(56+16\right)=14,4\left(g\right)\)
Câu 3 :
Cu không tác dụng với HCl, chỉ có Zn phản ứng.
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
Ta có: \(n_{H2}=\frac{4,48}{22,4}=0,2\left(mol\right)\)
Theo phản ứng: \(n_{Zn}=n_{H2}=0,2\left(mol\right)\rightarrow m_{Zn}=0,2.65=13\left(g\right)\)
\(\rightarrow\%m_{Zn}=\frac{13}{20}=65\%\rightarrow\%m_{Cu}=35\%\)
Ta có: \(n_{HCl}=2n_{H2}=0,2.2=0,4\left(mol\right)\)
\(\Rightarrow V_{HCl}=\frac{0,4}{2}=0,2\left(l\right)\)
Câu 4:
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(Al+3HCl\rightarrow AlCl_3+\frac{3}{2}H_2\)
Gọi số mol Fe là x; Al là y
\(\rightarrow56x+27y=22\)
Ta có: \(n_{H2}=n_{Fe}=1,5n_{Al}=x+1,5y=\frac{17,92}{22,4}=0,8\left(mol\right)\)
Giải được: \(\rightarrow\left\{{}\begin{matrix}x=0,2\\y=0,4\end{matrix}\right.\)
\(\Rightarrow m_{Fe}=0,2.56=11,2\left(g\right)\)
\(\rightarrow\%m_{Fe}=\frac{11,2}{22}=50,9\%\rightarrow\%m_{Al}=49,1\%\)
Ta có: \(n_{HCl}=2n_{H2}=1,6\left(mol\right)\)
\(\rightarrow m_{HCl}=1,6.36,5=58,4\left(g\right)\)
\(\rightarrow m_{dd_{HCl}}=\frac{58,4}{7,3\%}=800\left(g\right)\)
Câu 5:
Gọi chung 2 kim loại là R hóa trị I
\(R+HCl\rightarrow RCl+\frac{1}{2}H_2\)
Ta có: \(n_{H2}=\frac{0,448}{22,4}=0,02\left(mol\right)\rightarrow n_{RCl}=2n_{H2}=0,04\left(mol\right)\)
\(\rightarrow m_{RCl}=0,04.\left(R+35,5\right)=2,58\rightarrow R=29\)
Vì 2 kim loại liên tiếp nhau \(\rightarrow\) 2 kim loại là Na x mol và K y mol
\(\rightarrow x+y=n_{RCl}=0,04\left(mol\right)\)
\(m_{hh}=m_R=23x+39y=0,04.29=1,16\left(g\right)\)
Giải được: \(\rightarrow\left\{{}\begin{matrix}x=0,025\\y=0,015\end{matrix}\right.\)
\(\rightarrow m_{Na}=0,575\left(g\right)\)
\(\rightarrow\%m_{Na}=\frac{0,575}{1,16}=49,57\%\rightarrow\%m_K=50,43\%\)
Câu 6:
Khối lượng mỗi phần là 35/2=17,5g
Gọi số mol Fe, Cu, Al là a, b, c
Ta có \(56a+64b=27c=17,5\)
Phần 1: \(n_{H2}=\frac{6,72}{22,4}=0,3\left(mol\right)\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(\Rightarrow a=1,5b=n_{H2}=0,3\)
Phần 2: \(n_{Cl2}=\frac{10,64}{22,4}=0,475\left(mol\right)\)
\(2Fe+3Cl_2\rightarrow2FeCl_3\)
\(Cu+Cl_2\rightarrow CuCl_2\)
\(2Al+3Cl_2\rightarrow2AlCl_3\)
\(\Rightarrow1,5a+b+1,5c=n_{Cl2}=0,465\)
\(\rightarrow\left\{{}\begin{matrix}a=0,15\\b=0,1\\c=0,1\end{matrix}\right.\)
\(\rightarrow\%m_{Fe}=\frac{0,15.56}{17,5}=48\%\)
\(\rightarrow\%m_{Cu}=\frac{0,1.64}{17,5}=36,57\%\)
\(\rightarrow\%m_{Al}=100\%-48\%-36,57\%=15,43\%\)
Câu 1
2Al+6HCl--->2Alcl3+3H2
x-----------------------1,5x
Zn+2HCl---->Zncl2+H2
y---------------------------y
n H2=1/2=0,5(mol)
Theo bài ta có hpt
\(\left\{{}\begin{matrix}27x+65y=18,4\\1,5x+y=0,5\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}x=0,2\\y=0,2\end{matrix}\right.\)
%m Al=0,2.27/18,4.100%=29,35%
%m Zn=100%-29,35=70,65%
Câu 2.
Fe+2HCl---->FeCl2+H2
FeO+2HCl--->FeCl2+H2
n H2=2,24/22,4=0,1(mol)
m H2=0,2(g)
n Fe=n H2=0,2(mol)
m Fe=0,2.56=11,2(g)
n FeCl2(1)=2n H2=0,2(mol)
m FeCl2(1)=0,2.127=25,4(g)
m FeCl2(PT2)=38,1-25,4=12,7(g)
n FeCl2=12,7/127=0,1(mol)
n FeO=n FeCl2=0,1(mol)
m FeO=0,1.72=7,2(g)
3.
Zn+2HCl--->ZnCl2+H2
n H2=4,48/22,4=0,2(mol)
n Zn=n H2=0,2(mol)
m Zn=0,2.56=11,2(g)
%m Zn=11,2/20.100%=56%
%m Cu=100-56=34%
b) n HCl=2n H2=0,4(mol)
V H2=0,4/2=0,2(l)
4.
a) Fe+2HCl---.FeCl2+H2
x-----------------------------x(mol)
2Al+6HCl--->AlCl3+3H2
y------------------------------1,5y
n H2=17,92/22,4=0,89mol)
Theo bài ta có hpt
\(\left\{{}\begin{matrix}56x+27y=22\\x+1,5y=0,8\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}x=0,2\\y=0,4\end{matrix}\right.\)
%m Fe=0,2.56/22.100%=50,9%
%m Al=100-50,9=49,1%
b) n HCl=2n H2=1,6(mol)
m HCl=1,6.36,5=58,4(g)
m dd HCl=58,4.100/7,3=800(g)
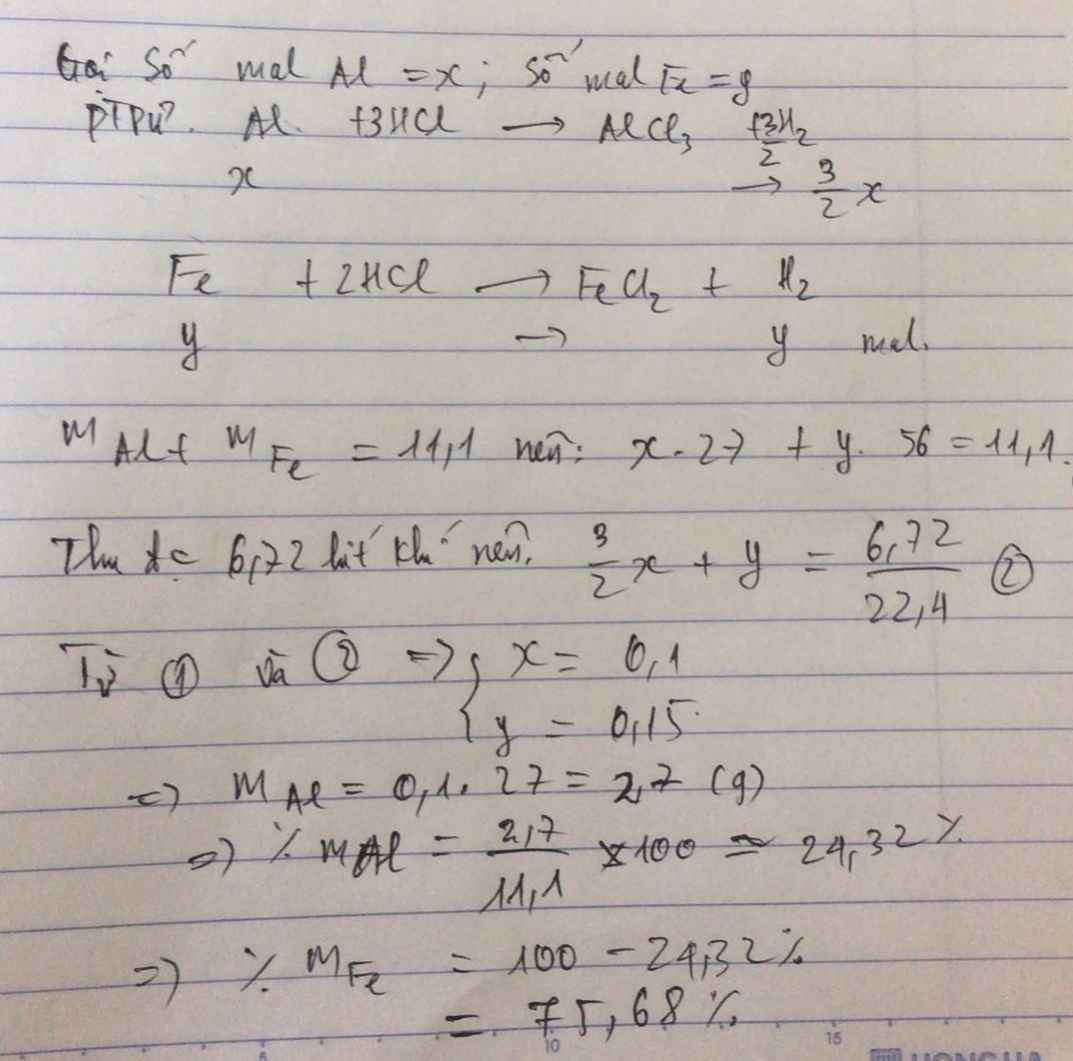




\(4.\)
\(n_{H_2}=\dfrac{3.36}{22.4}=0.15\left(mol\right)\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(0.15.....0.3....................0.15\)
\(m_{Fe}=0.15\cdot56=8.4\left(g\right)\)
\(C_{M_{HCl}}=\dfrac{0.3}{0.5}=0.6\left(M\right)\)
\(5.\)
\(Đặt:n_{Fe}=a\left(mol\right),n_{Al}=b\left(mol\right)\)
\(m_{hh}=56a+27b=8.3\left(g\right)\left(1\right)\)
\(n_{H_2}=\dfrac{5.6}{22.4}=0.25\left(mol\right)\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(\Rightarrow a+1.5b=0.25\left(2\right)\)
\(\left(1\right),\left(2\right):a=b=0.1\)
\(\%Fe=\dfrac{5.6}{8.3}\cdot100\%=67.47\%\)
\(\%Al=32.53\%\)
bạn ơi cho mik hỏi: tại sao lại suy ra: a+1,5b=0,25 vậy ạ ? và cả bước tiếp theo nx ạ ?