Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) Gọi số mol Zn, Fe là a, b (mol)
=> 65a + 56b = 8,56 (1)
\(n_{H_2}=\dfrac{3,136}{22,4}=0,14\left(mol\right)\)
PTHH: Zn + 2HCl --> ZnCl2 + H2
a--->2a-------->a----->a
Fe + 2HCl --> FeCl2 + H2
b----->2b------->b------>b
=> a + b = 0,14 (2)
(1)(2) => a = 0,08; b = 0,06
=> \(\left\{{}\begin{matrix}\%m_{Zn}=\dfrac{0,08.65}{8,56}.100\%=60,748\%\\\%m_{Fe}=\dfrac{0,06.56}{8,56}.100\%=39,252\%\end{matrix}\right.\)
b)
nKOH = 0,2.0,1 = 0,02 (mol)
PTHH: KOH + HCl --> KCl + H2O
0,02-->0,02
=> nHCl = 0,02 + 2a + 2b = 0,3 (mol)
=> \(C_{M\left(HCl\right)}=xM=\dfrac{0,3}{0,15}=2M\)
c) m = 0,08.136 + 0,06.127 = 18,5(g)

a) Gọi số mol Mg, Fe là a, b (mol)
=> 24a + 56b = 11,84
\(n_{HCl}=\dfrac{146.14\%}{36,5}=0,56\left(mol\right)\)
PTHH: Mg + 2HCl --> MgCl2 + H2
a--->2a--------->a----->a
Fe + 2HCl --> FeCl2 + H2
b-->2b-------->b------>b
=> 2a + 2b = 0,56
=> a = 0,12; b = 0,16
=> \(\left\{{}\begin{matrix}\%Mg=\dfrac{0,12.24}{11,84}.100\%=24,324\%\\\%Fe=\dfrac{0,16.56}{11,84}.100\%=75,676\%\end{matrix}\right.\)
b) \(n_{H_2}=a+b=0,28\left(mol\right)\)
=> \(V_{H_2}=0,28.22,4=6,272\left(l\right)\)
c) mdd sau pư = 11,84 + 146 - 0,28.2 = 157,28 (g)
=> \(\left\{{}\begin{matrix}C\%_{MgCl_2}=\dfrac{0,12.95}{157,28}.100\%=7,25\%\\C\%_{FeCl_2}=\dfrac{0,16.127}{157,28}.100\%=12,92\%\end{matrix}\right.\)

Giả sử: \(\left\{{}\begin{matrix}n_{Mg}=x\left(mol\right)\\n_{Al}=y\left(mol\right)\end{matrix}\right.\)
⇒ 24x + 27y = 7,8 (1)
Ta có: \(n_{H_2}=\dfrac{8,96}{22,4}=0,4\left(mol\right)\)
BT e, có: 2x + 3y = 0,8 (2)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}x=0,1\left(mol\right)\\y=0,2\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}m_{Mg}=0,1.24=2,4\left(g\right)\\m_{Al}=0,2.27=5,4\left(g\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{Mg}=\dfrac{2,4}{7,8}.100\%\approx30,77\%\\\%m_{Al}\approx69,23\%\end{matrix}\right.\)
b, BTNT Mg và Al, có:
nMgCl2 = nMg = 0,1 (mol)
nAlCl3 = nAl = 0,2 (mol)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{MgCl_2}=\dfrac{0,1.95}{0,1.95+0,2.133,5}.100\%\approx26,24\%\\\%m_{AlCl_3}\approx73,76\%\end{matrix}\right.\)
Bạn tham khảo nhé!

a) \(n_{AlCl_3}=\dfrac{6,675}{133,5}=0,05\left(mol\right)\)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
0,05<-----------0,05---->0,075
=> \(\%Al=\dfrac{0,05.27}{14,15}.100\%=9,54\%\)
=> \(\%Cu=\dfrac{14,15-0,05.27}{14,15}.100\%=90,46\%\)
b) \(V_{H_2}=0,075.22,4=1,68\left(l\right)\)
c) \(n_{Cu}=\dfrac{14,15-0,05.27}{64}=0,2\left(mol\right)\)
PTHH: 4Al + 3O2 --to--> 2Al2O3
0,05->0,0375
2Cu + O2 --to--> 2CuO
0,2-->0,1
=> \(V_{O_2}=\left(0,1+0,0375\right).22,4=3,08\left(l\right)\)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\\ m_{AlCl_3}=6,675\left(mol\right)\\ n_{AlCl_3}=\dfrac{6,675}{133,5}=0,05\left(mol\right)\\ \Rightarrow n_{Al}=n_{AlCl_3}=0,05\left(mol\right)\\ \Rightarrow m_A=0,05.27=1,35\left(g\right);m_{Cu}=14,15-1,35=12,8\left(g\right)\\ \%m_{Cu}=\dfrac{12,8}{14,15}.100\approx90,459\%\\ \Rightarrow\%m_{Al}\approx9,541\%\\ b,n_{Cu}=\dfrac{12,8}{64}=0,2\left(mol\right)\\ n_{H_2}=\dfrac{3}{2}.n_{Al}=\dfrac{3}{2}.0,05=0,075\left(mol\right)\\ \Rightarrow V=V_{H_2\left(đktc\right)}=0,075.22,4=1,68\left(l\right)\\ 4Al+3O_2\rightarrow\left(t^o\right)2Al_2O_3\\ 2Cu+O_2\rightarrow\left(t^o\right)2CuO\\ n_{O_2}=\dfrac{3}{4}.n_{Al}+\dfrac{1}{2}.n_{Cu}=\dfrac{3}{4}.0,05+\dfrac{1}{2}.0,2=0,0875\left(mol\right)\)
\(\Rightarrow V_{O_2\left(đktc\right)}=0,0875.22,4=1,96\left(l\right)\)

Gọi số mol Al, Mg là a, b (mol)
\(n_{H_2}=\dfrac{13,44}{22,4}=0,6\left(mol\right)\)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
a--------------->a------>1,5a
Mg + 2HCl --> MgCl2 + H2
b--------------->b---->b
=> \(\left\{{}\begin{matrix}1,5a+b=0,6\\133,5a+95b=55,2\end{matrix}\right.\)
=> a = 0,2 (mol); b = 0,3 (mol)
\(\left\{{}\begin{matrix}\%m_{Al}=\dfrac{0,2.27}{0,2.27+0,3.24}.100\%=42,857\%\\\%m_{Mg}=\dfrac{0,3.24}{0,2.27+0,3.24}.100\%=57,143\%\end{matrix}\right.\)
\(\left\{{}\begin{matrix}Al\\Mg\end{matrix}\right.+HCl->\left\{{}\begin{matrix}AlCl3\\MgCl2\end{matrix}\right.+H2\)
2Al + 3HCl -> 2AlCl3 + 3H2
0,2 0,3 0,3
Mg + 2HCl -> MgCl2 + H2
0,3 0,6 0,3
=> mHCl dùng = 0,9 . 36,5 = 32,85 (g)
=> mH2 = 0,6 . 2 = 1,2 (g)
Bảo toàn khối lượng :
=> mX = 55,2 + 1,2 - 32,85 = 23,55 (g)
Ta có :
\(\left\{{}\begin{matrix}3x+2y=1,2\left(bt-e\right)\\133,5x+95y=55,2\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}x=0,2\left(mol\right)\\y=0,3\left(mol\right)\end{matrix}\right.\)
\(\left\{{}\begin{matrix}\%mAl=\dfrac{0,2.27}{0,2.27+0,3.24}=42,85\%\\\%mMg=100\%-42,85\%=57,15\%\end{matrix}\right.\)


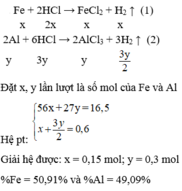
Câu 4:
\(n_{H2}=\frac{13,44}{22,4}=0,6\left(mol\right)\)
Đặt trong 22,2 gam hh Al và Fe số mol mỗi chất lần lượt là x và y (mol)
\(\Rightarrow m_{hh}=m_{Al}+m_{Fe}\)
\(\Rightarrow27x+56y=22,2\left(I\right)\)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\left(1\right)\)
x____________________1,5x
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
y_____________________y
Theo PTHH (1):
\(n_{H2\left(1\right)}=\frac{3}{2}n_{Al}=\frac{3}{2}.x=1,5x\left(mol\right)\)
Theo PTHH (2):
\(n_{H2\left(2\right)}=n_{Fe}=y\left(mol\right)\)
\(\Rightarrow\Sigma n_{H2}=n_{H2\left(1\right)}+n_{H2\left(2\right)}\)
\(\Rightarrow1,5x+y=0,6\left(II\right)\)
Giải hệ (I) và (II) ta được \(\left\{{}\begin{matrix}x=0,2\\y=0,3\end{matrix}\right.\)
\(m_{Al}=0,2.27=5,4\left(g\right)\)
\(\%m_{Al}=\frac{5,4}{22,2}.100\%=23,32\%\)
\(\%m_{Fe}=100\%-23,32\%=76,68\%\)
Câu 5:
a, \(n_{H2}=\frac{12,32}{22,4}=0,55\left(mol\right)\)
Gọi số mol Fe, Zn lần lượt là a;b
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
Ta có phương trình:
\(\left\{{}\begin{matrix}56a+65b=32,6\\a+b=0,55\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}a=0,35\left(mol\right)\\b=0,2\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\%m_{Fe}=\frac{19,6}{21,6}.100\%=60,12\%\)
\(\Rightarrow\%m_{Zn}=100\%-60,12\%=39,88\%\)
b,\(n_{Fe}=n_{FeCl2}=0,35\left(mol\right)\)
\(\Rightarrow m_{FeCl2}=0,35.127=44,45\left(g\right)\)
\(n_{ZnCL2}=n_{Zn}=0,2\left(mol\right)\)
\(\Rightarrow m_{ZnCl2}=0,2.136=27,2\left(g\right)\)
Bài 4. Cho 22,2 gam hỗn hợp gồm Al và Fe vào dung dịch HCl loãng dư, thu được dung dịch X và 13,44 lít khí thoát ra (ở điều kiện chuẩn).
a/ Tính % khối lượng mỗi kim loại trong hỗn hợp.
b/ Tính khối lượng mỗi muối trong dung dịch X?
-------------------------Giải--------------------
Đặt x_n Fe ; y_n Al
PTHH:
\(Fe+2HCl\rightarrow FeCl_2+H_2\uparrow\) (1)
x..........................................x
\(2Al+6HCl\rightarrow2AlCl_2+3H_2\uparrow\)(2)
y..............................................\(\frac{3}{2}y\)
\(n_{H_2}=\frac{13,44}{22,4}=0,6\left(mol\right)\)
Ta có : \(\left\{{}\begin{matrix}x+\frac{3}{2}y=0,6\\56x+27y=22,2\end{matrix}\right.\)\(\Leftrightarrow\left\{{}\begin{matrix}x=0,3\\y=0,2\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}m_{Fe}=0,3.56=16,8\left(g\right)\\m_{Al}=0,2.27=5,4\left(g\right)\end{matrix}\right.\)
\(\Rightarrow\%m_{Fe}=\frac{16,8}{22,2}.100=75,68\%\)
\(\Rightarrow\%m_{Al}=100-75,68=24,32\%\)
b) Theo PT (1) : \(n_{FeCl_2}=n_{Fe}=0,3\left(mol\right)\)
\(\Rightarrow m_{FeCl_2}=0,3.127=38,1\left(g\right)\)
Theo PT (2): \(n_{AlCl_3}=n_{Al}=0,2\left(mol\right)\)
\(\Rightarrow m_{AlCl_3}=0,2.133,5=26,7\left(g\right)\)