Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a)
C% CuSO4 = 16/(16 + 184) .100% = 8%
b)
n NaOH = 20/40 = 0,5(mol)
CM NaOH = 0,5/4 = 0,125M

a, \(C\%_{NaOH}=\dfrac{4}{4+2,8+118,2}.100\%=3,2\%\)
\(C\%_{KOH}=\dfrac{2,8}{4+2,8+118,2}.100\%=2,24\%\)
b, \(n_{NaOH}=\dfrac{4}{40}=0,1\left(mol\right)\)
\(\Rightarrow C_{M_{NaOH}}=\dfrac{0,1}{0,125}=0,8\left(M\right)\)
\(n_{KOH}=\dfrac{2,8}{56}=0,05\left(mol\right)\)
\(\Rightarrow C_{M_{KOH}}=\dfrac{0,05}{0,125}=0,4\left(M\right)\)

a.\(n_{NaOH}=\dfrac{8}{40}=0,2mol\)
\(V_{dd}=\dfrac{120}{1,2}=100ml=0,1l\)
\(C_{M_{NaOH}}=\dfrac{0,2}{0,1}=2M\)
b.\(n_{NaOH}=\dfrac{21,6}{40}=0,54mol\)
\(V_{dd}=\dfrac{180}{1,2}=150ml=0,15l\)
\(C_{M_{NaOH}}=\dfrac{0,54}{0,15}=3,6M\)

\(a,C\%_{KOH}=\dfrac{28}{140}.100\%=20\%\\ b,C\%_{KOH}=\dfrac{80}{80+320}.100\%=20\%\)

a, \(C\%_{KCl}=\dfrac{20}{20+60}.100\%=25\%\)
b, \(C\%=\dfrac{40}{40+150}.100\%\approx21,05\%\)
c, \(C\%_{NaOH}=\dfrac{60}{60+240}.100\%=20\%\)
d, \(C\%_{NaNO_3}=\dfrac{30}{30+90}.100\%=25\%\)
e, \(m_{NaCl}=150.60\%=90\left(g\right)\)
f, \(m_{ddA}=\dfrac{25}{10\%}=250\left(g\right)\)
g, \(n_{NaOH}=120.20\%=24\left(g\right)\)
Gọi: nNaOH (thêm vào) = a (g)
\(\Rightarrow\dfrac{a+24}{a+120}.100\%=25\%\Rightarrow a=8\left(g\right)\)

\(n_{Na_2O}=\dfrac{6,2}{62}=0,1mol\\ Na_2O+H_2O\rightarrow2NaOH\\ n_{NaOH}=0,1.2=0,2mol\\ C_{M_X}=C_{M_{NaOH}}=\dfrac{0,2}{2}=0,1M\)
\(n_{Na_2O}=\dfrac{6,2}{62}=0,1\left(mol\right)\)
PT: \(Na_2O+H_2O\rightarrow2NaOH\)
Theo PT: \(n_{NaOH}=2n_{Na_2O}=0,2\left(mol\right)\)
\(\Rightarrow C_{M_{NaOH}}=\dfrac{0,2}{2}=0,1\left(m\right)\)

\(a)m_{dd}=4+2,8+118,2=125g\\ C_{\%NaOH}=\dfrac{4}{125}\cdot100\%=3,2\%\\ C_{\%KOH}=\dfrac{2,8}{125}\cdot100\%=2,24\%\\ b)n_{NaOH}=\dfrac{4}{40}=0,1mol\\ \\ n_{KOH}=\dfrac{2,8}{56}=0,05mol\\ 125ml=0,125l\\ C_{M_{NaOH}}=\dfrac{0,1}{0,125}=0,8M\\ C_{M_{KOH}}=\dfrac{0,05}{0,125}=0,4M\)
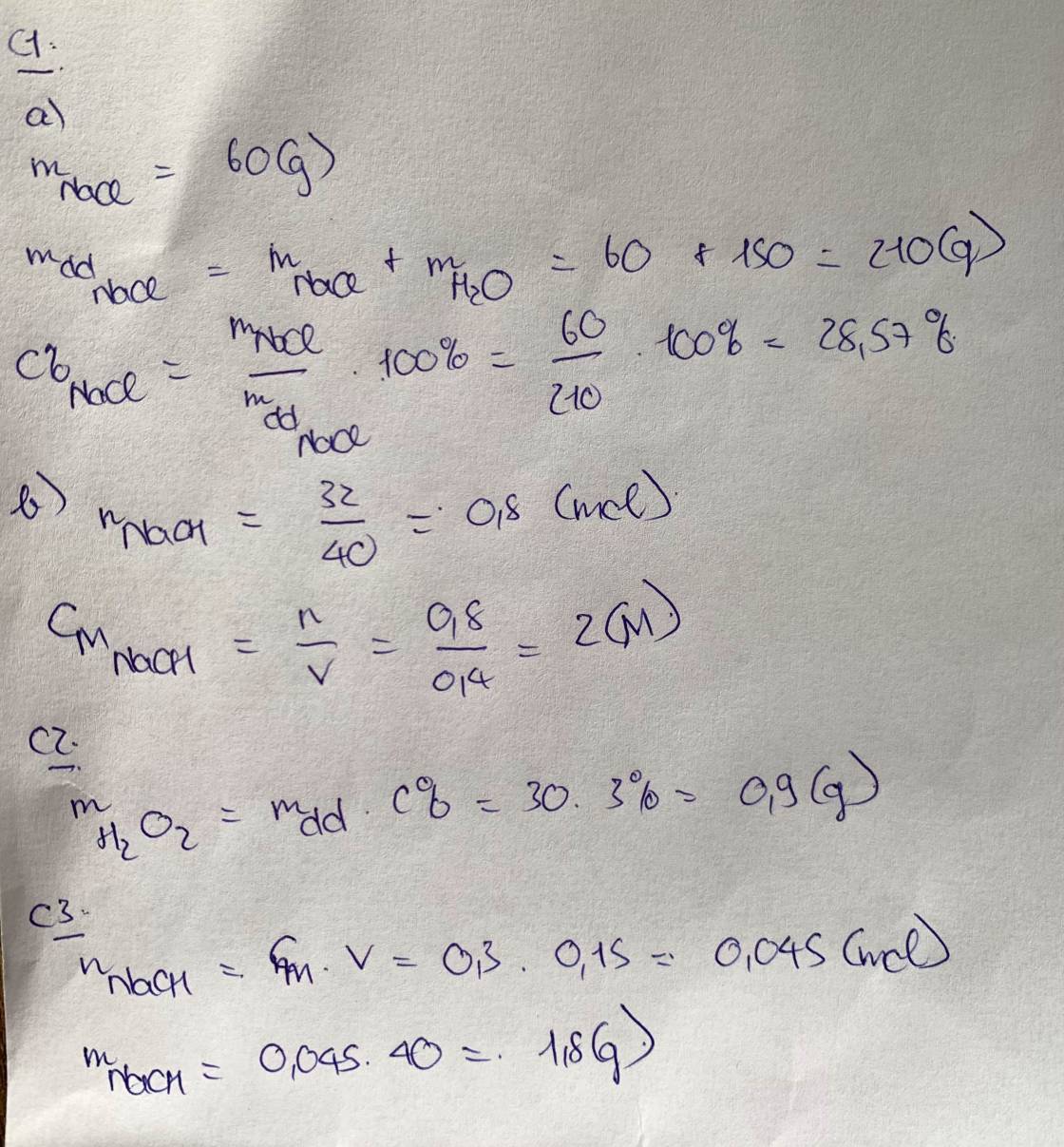
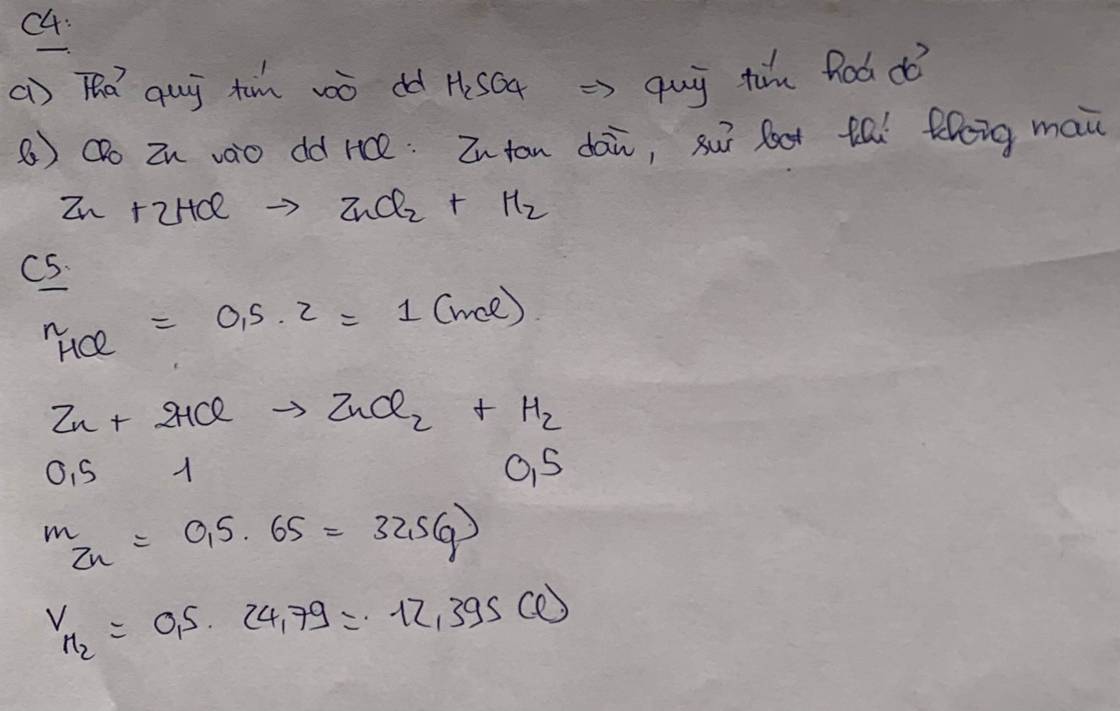
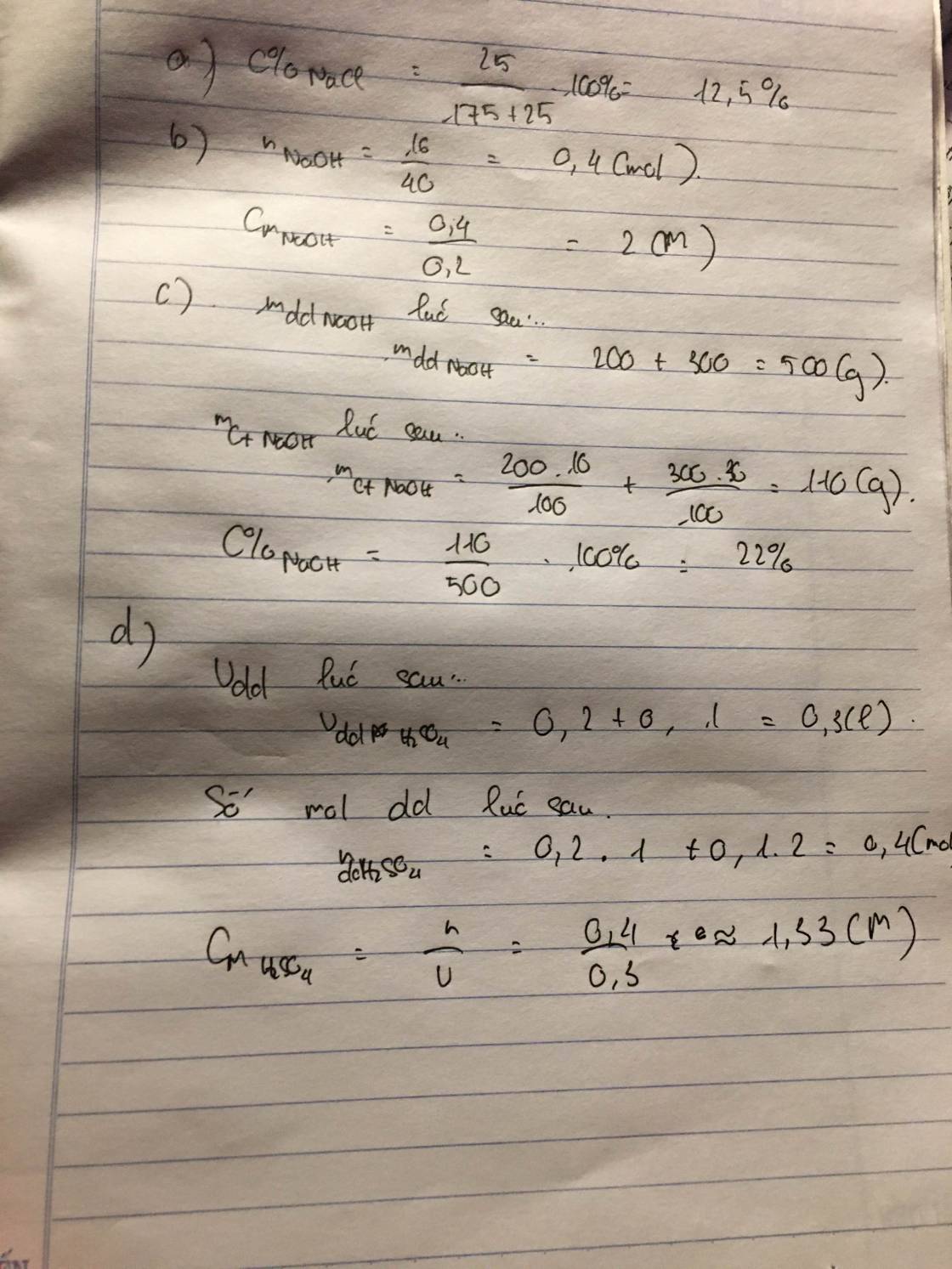
\(a,C_{M\left(NaOH\right)}=\dfrac{0,3}{0,5}=0,6M\\ b,n_{NaOH}=\dfrac{24}{40}=0,6\left(mol\right)\\ C_{M\left(NaOH\right)}=\dfrac{0,6}{0,4}=1,5M\)