Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) Axit sunfuric + kẽm oxit → Kẽm sunfat + Nước
H2SO4 + ZnO → ZnSO4 + H2O
b) Natri hiđroxit + lưu huỳnh trioxit → Natri sunfat + Nước
2NaOH + SO3 → Na2SO4 + H2O
c) Nước + lưu huỳnh đioxit → Axit sunfurơ
H2O + SO2 → H2SO3
d) Nước + canxi oxit → Canxi hiđroxit
H2O + CaO → Ca(OH)2
e) Canxi oxit + cacbon đioxit→ Canxi cacbonat
CaO + CO2 → CaCO3

a) H2SO4 + ZnO → ZnSO4 + H2O
b) 2NaOH + SO3 → Na2SO4 + H2O
c) H2O + SO2 → H2SO3
d) H2O + CaO → Ca(OH)2
e) CaO + CO2 → CaCO3

a) kẽm oxit
b) lưu huỳnh trioxit
c)lưu huỳnh đioxit
d)canxi oxit
e)cacbon đioxit
PTHH :
a)H2SO4+ZnO->ZnSO4+H2O
B)NaOH+SO3->NaSO4+H2O
c)H2O+SO2->H2SO3
d)H2O+CaO->Ca(OH)2
e)CaO+CO2->CaCO3

1, CO2+H2O--->H2CO3
2, SO3+H2O--->H2SO4
3, SO2+H2O--->H2SO3
4, N2O5+H2O---> 2HNO3
5, P2O5+3H2O--->2H3PO4
6, SO2+K2O--->K2SO3
7, CO2+BaO--->BaCO3
8, SO3+Na2O--->Na2SO4
9, P2O5+3CaO--->Ca3(PO4)2
10, N2O5+K2O--->2KNO3
11, CO2+2NaOH--->Na2CO3+H2O
12, SO2+Ca(OH)2--->CaSO3+H2O
13, SO3+Ba(OH)2--->BaSO4+H2O
14, N2O5+2KOH--->2KNO3+H2O
15, P2O5+3Ba(OH)2--->Ba3(PO4)2+3H2O
16, Na2O+H2O--->2NaOH
17, K2O+H2O--->2KOH
18, CaO+H2O--->Ca(OH)2
19, BaO+H2O--->Ba(OH)2
20, Al2O3+6HCl--->2AlCl3+3H2O
21, Fe2O3+6HNO3---> 2Fe(NO3)3+3H2O
22, ZnO+H2SO4--->ZnSO4+H2O
23, 3CaO+2H3PO4--->Ca3(PO4)2+3H2O
24, Fe+2HCl--->FeCl2+H2
25, Mg+H2SO4--->MgSO4+H2
26, 2Al+6HCl--->2AlCl3+3H2
27, Zn+H2SO4---->ZnSO4+H2
28, Cu+2H2SO4---> CuSO4+SO2+2H2O
29, Al(OH)3+3HCl--->AlCl3+3H2O
30, Zn(OH)2+H2SO4--->ZnSO4+2H2O

1.
Na2O + SO2\(\rightarrow\)Na2SO3
2.
CaO + H2O \(\rightarrow\)Ca(OH)2
3.ko xảy ra
4.
Al2O3 + 6HNO3 \(\rightarrow\)2Al(NO3)3 + 3H2O
5.
2Fe(OH)3 + 3H2SO4 \(\rightarrow\)Fe2(SO4)3 + 6H2O
6.
ZnO + SO3 \(\rightarrow\)ZnSO4
7.
Fe + H2SO4 \(\rightarrow\)FeSO4 + H2
8.ko xảy ra
9.
CuO + 2HNO3 \(\rightarrow\)Cu(NO3)2 + H2O
10.
2NaOH + H2SO4 \(\rightarrow\)Na2SO4 + 2H2O
11.
Fe(OH)3 + 3HNO3 \(\rightarrow\)Fe(NO3)3 + 3H2O
12.
Cu(OH)2 + 2HCl \(\rightarrow\)CuCl2 + 2H2O

a) Sơ đồ phản ứng:
S → SO2 → SO3 → H2SO4 → Na2SO4 → BaSO4.
b) Phương trình phản ứng:

\(a.Zn+H_2SO_4\rightarrow ZnSO_4+H_2\\ b.Zn+2AgNO_3\rightarrow Zn\left(NO_3\right)_2+2Ag\\ c.2Na+S\xrightarrow[]{t^0}Na_2S\\ d.Ca+Cl_2\xrightarrow[]{t^0}CaCl_2\\ e.MgO+2HNO_3\rightarrow Mg\left(NO_3\right)_2+H_2O\\ f.Fe+2HCl\rightarrow FeCl_2+H_2\\ g.CuO+2HCl\rightarrow CuCl_2+H_2O\\ h.2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\\ i.Cl+2NaOH\rightarrow NaCl+NaClO+H_2O\\ k.MgO_2+4HCl_{đặc}\xrightarrow[nhẹ]{đun}MgCl_2+Cl_2+2H_2O\)

a) H2O+SO3-->H2SO4
b) CO2+H2O--->H2CO3
c) 3H2O+P2O5--->2H3PO4
d) CaO+H2O--->Ca(OH)2
e) Na2O+H2O--->2NaOH
\(a,\text{H2O + SO3 → H2SO4}\)
\(b,\text{H2O + CO2 ↔ H2CO3}\)
\(c,\text{3H2O + P2O5 → 2H3PO4}\)
\(d,\text{CaO + H2O → Ca(OH)2}\)
\(e,\text{H2O + Na2O → 2NaOH}\)

Phương trình hóa học:
a) H2 + F2 → 2HF (k)
b) S + O2 → SO2(to)
c) Fe + S → FeS (to).
d) C + O2 → CO2(to).
e) H2 + S → H2S.(to)
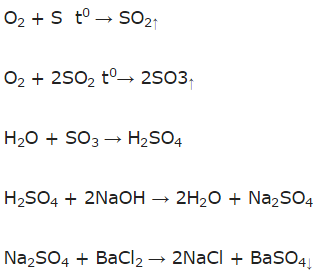
a) H2SO4 + ZnO → ZnSO4 + H2O
b) 2NaOH + SO3 → Na2SO4 + H2O
c) H2O + SO2 → H2SO3
d) H2O + CaO → Ca(OH)2
e) CaO + CO2 → CaCO3
a) Axit sunfuric + Kem oxit \(\rightarrow\) Kẽm sunfat + Nuoc
b) Natri hidroxit + axit sunfuric \(\rightarrow\) Natri sunfat + nuoc
c) Nước + Lưu huỳnh dioxit \(\rightarrow\) Axit sunfuro
d) Nước + canxi oxit \(\rightarrow\) Canxi hidroxit
e) Canxi oxit + cacbon dioxit \(\rightarrow\) Canxi cacbonat